How can chemistry be considered the blueprint of matter?
Earth and Atmospheric Sciences
The Science of Heat and Chemical Change
Thermochemistry is the branch of physical chemistry that studies the energy and heat associated with chemical reactions and physical transformations (such as melting or boiling). It focuses on the energy exchange between a system (the substances undergoing change) and its surroundings (everything else).
At its core, thermochemistry helps us understand why some reactions release energy (like a burning candle) while others absorb it (like a cold pack).
Key Concepts in Thermochemistry
To understand how energy moves during a reaction, we use several fundamental terms and laws:
- Enthalpy (H): This is the total heat content of a system. In chemistry, we are usually interested in the change in enthalpy (ΔH), which is the heat absorbed or released during a reaction at constant pressure.
- Exothermic Reactions: These reactions release heat into the surroundings. Because the system loses energy, the change in enthalpy is negative (-ΔH).
- Example: Combustion (burning fuel) or freezing water.
- Endothermic Reactions: These reactions absorb heat from the surroundings. The system gains energy, resulting in a positive change in enthalpy (+ΔH).
- Example: Photosynthesis or melting ice.
- Calorimetry: This is the experimental process of measuring the heat of chemical reactions or physical changes using an instrument called a calorimeter.
Governing Laws
Thermochemistry is built upon the laws of thermodynamics, specifically:
- First Law of Thermodynamics (Law of Conservation of Energy): Energy cannot be created or destroyed, only transferred or transformed. In a reaction, the energy lost by the system must equal the energy gained by the surroundings.
- Hess’s Law: This law states that the total enthalpy change for a chemical reaction is the same regardless of whether the reaction occurs in one step or several steps. This allows scientists to calculate $\Delta H$ for complex reactions by breaking them down into simpler, known steps.
Why It Matters
Thermochemistry is more than just equations; it has vital practical applications:
- Fuel Efficiency: Calculating how much energy is produced by different types of coal, gas, or biofuels.
- Material Science: Designing materials that can withstand high heat or phase changes.
- Biology: Understanding how our bodies break down food (metabolism) to produce the energy required for life.
- Weather and Climate: Studying how the phase changes of water (evaporation and condensation) drive atmospheric energy and weather patterns.
How does some terminology get us started in thermochemistry?
To understand how energy moves during a chemical change, we have to define exactly what we are looking at and where the energy is going. Thermochemistry uses a specific set of terms to create a “map” of these energy transfers.
1. The System and the Surroundings
In any experiment, we divide the entire universe into two parts to keep track of energy flow:
- The System: This is the specific part of the universe we are studying. In a lab, it is usually the chemicals inside a beaker or a flask.
- The Surroundings: This is everything else outside the system—the beaker itself, the air in the room, and even the scientist performing the experiment.
There are three main types of systems based on how they interact with their surroundings:
- Open System: Can exchange both mass and energy (like an open cup of hot coffee).
- Closed System: Can exchange energy but not mass (like a coffee cup with a tight lid).
- Isolated System: Cannot exchange mass or energy (like a high-quality vacuum thermos).
2. State Functions
A state function is a property that depends only on the current state of the system (like its temperature, pressure, or volume), not on how it got there.
Think of it like climbing a mountain: your altitude is a state function (it’s the same whether you took the steep path or the winding trail), but the distance you walked is not. In thermochemistry, Enthalpy (H) and Internal Energy (E) are state functions.
3. Direction of Heat Flow: Exo vs. Endo
The prefix tells you the direction the “heat” (thermos) is moving:
- Exothermic (Exo = “Exit”): Heat flows out of the system and into the surroundings. The surroundings get hotter.
- Sign: ΔH < 0 (Negative).
- Endothermic (Endo = “Inside”): Heat flows into the system from the surroundings. The surroundings get colder.
- Sign: ΔH > 0 (Positive).
4. Enthalpy (H) and Change in Enthalpy (ΔH)
While total enthalpy is hard to measure directly, we can easily measure the change (Δ) in enthalpy. This represents the heat flow at constant pressure.
ΔH = Hproducts – Hreactants
- If the products have less energy than the reactants, the extra energy was released (Exothermic).
- If the products have more energy, the system must have absorbed it (Endothermic).
5. Specific Heat Capacity (c)
Not every substance heats up at the same rate. Specific heat is the amount of heat required to raise the temperature of 1 gram of a substance by 1 degree Celsius (or 1 Kelvin).
- Water has a very high specific heat (4.184 J/g°C), which is why it takes a long time to boil but also stays warm for a long time.
- Metals usually have low specific heats, meaning they heat up and cool down very quickly.
What is work?
In the context of thermodynamics and thermochemistry, work (w) is defined as the transfer of energy that occurs when a force acts on an object over a distance.
While heat is the transfer of energy due to a temperature difference, work is the transfer of energy through organized motion. In chemistry, we most commonly deal with Pressure-Volume (PV) work.
1. Pressure-Volume (PV) Work
In a chemical system, work is usually done by or on a gas. Imagine a gas trapped inside a cylinder with a movable piston.
- Expansion: If the gas inside the cylinder expands, it pushes the piston upward against the external pressure. The system is doing work on the surroundings.
- Compression: If an external force pushes the piston down, the surroundings are doing work on the system.
2. The Mathematical Definition
The work done by a system during a volume change at constant external pressure (P) is calculated using the formula:
w = -PΔV
Where:
- P is the external pressure.
- ΔV is the change in volume (Vfinal – Vinitial).
Why the negative sign?
This follows the sign convention of thermodynamics. If a gas expands (ΔV is positive), the system loses energy by doing work on the surroundings. The negative sign ensures that the value for w is negative, reflecting that energy is leaving the system.
3. Work and Internal Energy
Work is one of the two primary ways to change the Internal Energy (ΔE or ΔU) of a system. This is expressed in the First Law of Thermodynamics:
ΔE = q + w
- q (Heat): Energy transfer via temperature difference.
- w (Work): Energy transfer via displacement or volume change.
4. Comparison of Sign Conventions
Understanding the “direction” of energy is critical in thermochemistry. You can think of the system like a bank account:
| Action | Process | Sign of Work (w) |
| Work done ON the system | Compression (Surroundings push in) | + (Energy added) |
| Work done BY the system | Expansion (System pushes out) | – (Energy spent) |
5. Work is Not a State Function
Unlike Enthalpy (H) or Temperature (T), work is a path function. This means the amount of work done depends on the specific steps taken to get from the start to the finish.
For example, if you compress a gas very quickly versus very slowly in multiple small stages, the total work performed will be different, even if the starting and ending pressures and volumes are identical.
What is heat?
In thermochemistry, heat (q) is the transfer of thermal energy between two bodies that are at different temperatures. It is important to distinguish that an object does not “contain” heat; rather, it contains internal energy. Heat only exists when that energy is in transit.
Thermal energy always flows spontaneously from a region of higher temperature to a region of lower temperature until thermal equilibrium is reached (when both objects are the same temperature).
1. Heat vs. Temperature
While they are related, they are not the same thing:
- Temperature (T): A measure of the average kinetic energy of the particles in a substance. It tells you how “hot” or “cold” something is.
- Heat (q): The total energy moved due to that temperature difference.
2. Direction of Heat Flow
In a chemical system, we track heat based on whether it enters or leaves the system:
- Positive (+q): Heat is absorbed by the system from the surroundings (Endothermic).
- Negative (-q): Heat is released by the system into the surroundings (Exothermic).
3. Calculating Heat (q)
To measure how much heat is transferred during a process, we use the heat capacity of the substance. The most common formula used in calorimetry is:
q = m * c * ΔT
Where:
- m: Mass of the substance (usually in grams).
- c: Specific heat capacity (the energy needed to raise 1g by 1°C).
- ΔT: The change in temperature (Tfinal – Tinitial).
4. Heat and Phase Changes
Heat doesn’t always change the temperature of a substance. When a substance reaches its melting or boiling point, the added heat is used to break the intermolecular forces holding the molecules together rather than increasing their speed.
- Latent Heat of Fusion: Heat required to turn a solid into a liquid.
- Latent Heat of Vaporization: Heat required to turn a liquid into a gas.
During these transitions, the temperature remains constant even though heat is still being added.
5. Methods of Heat Transfer
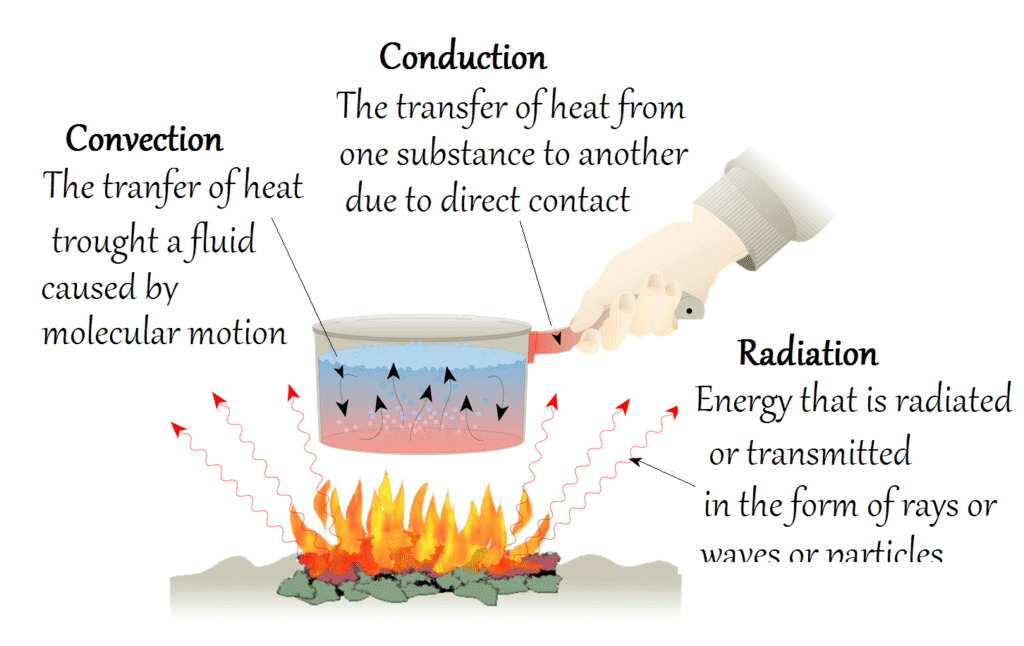
In a broader physical sense, heat moves in three ways:
- Conduction: Transfer through direct contact (e.g., a metal spoon in hot soup).
- Convection: Transfer through the movement of fluids or gases (e.g., warm air rising in the atmosphere).
- Radiation: Transfer through electromagnetic waves (e.g., energy from the sun reaching Earth).
What are heats of reaction and what is calorimetry?
While heat is the general transfer of energy, the heat of reaction and calorimetry are the specific tools chemists use to quantify exactly how much energy is “locked” inside chemical bonds.
1. Heats of Reaction (ΔHrxn)
The heat of reaction (also known as the Enthalpy of Reaction) is the amount of heat absorbed or released during a chemical reaction that takes place at constant pressure.
It is the difference between the enthalpy of the products and the enthalpy of the reactants:

- If ΔH is negative: The reaction is exothermic. The products have lower potential energy than the reactants, and the “leftover” energy is released as heat.
- If ΔH is positive: The reaction is endothermic. The products have higher potential energy, meaning energy had to be pulled from the surroundings to “climb” to that state.
2. Calorimetry: Measuring the Heat
Calorimetry is the experimental technique used to measure the heat exchange of a system. Since we cannot measure “enthalpy” directly, we measure the temperature change of the surroundings (usually water) and calculate the energy from there.
The device used for this is a calorimeter. There are two main types used in labs:
A. Constant-Pressure Calorimetry (Coffee-Cup Calorimeter)
Commonly used in introductory chemistry, this involves a simple insulated cup (like Styrofoam) containing a known mass of water. When a reaction happens in the water:
- The chemicals are the system.
- The water is the surroundings.
- We measure ΔT of the water and use q = mcΔT to find the heat.
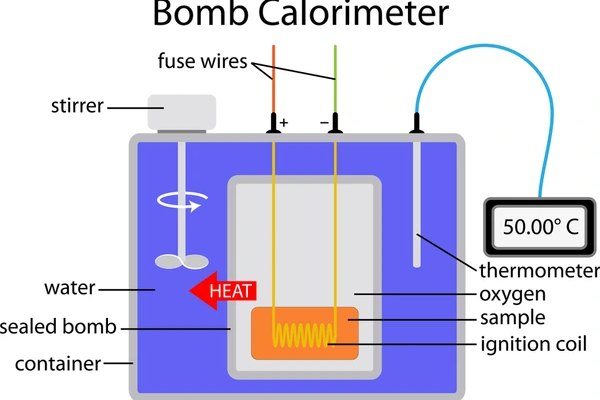
B. Constant-Volume Calorimetry (Bomb Calorimeter)
This is used for reactions that involve gases or high-energy combustion. The reaction happens inside a rigid, sealed “bomb” submerged in water. Because the volume cannot change, this measures the change in internal energy (ΔE) rather than enthalpy.
3. The Basic Calculation
To find the heat of a reaction in a simple calorimeter, we assume that all heat lost by the reaction (qrxn) is gained by the water (qsoln):
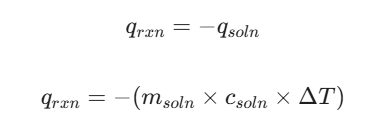
By dividing this heat (q) by the number of moles of the reactant used, you get the Molar Enthalpy of Reaction in kJ/mol.
4. Real-World Connection
This is how scientists determine the “Calories” in the food we eat. A sample of food is burned in a bomb calorimeter, and the resulting rise in water temperature tells us exactly how much chemical energy was stored in those fats, proteins, or carbohydrates.
What is the First Law of Thermodynamics?
The First Law of Thermodynamics is essentially the Law of Conservation of Energy, adapted for thermodynamic systems. It states that energy can be transformed from one form to another or transferred between a system and its surroundings, but it cannot be created or destroyed.
The total energy of the universe is constant.
1. The Mathematical Equation
In chemistry and physics, we express this law by looking at the change in the Internal Energy (ΔE or ΔU) of a system. Internal energy is the sum of all kinetic and potential energies of the particles inside the system.
ΔE = q + w
- ΔE: The change in internal energy.
- q: The heat exchanged between the system and surroundings.
- w: The work done on or by the system.
2. Understanding the Sign Convention
To apply the First Law correctly, you have to look at energy from the “perspective” of the system. Imagine the system is a bank account:
- Energy In (+q or +w): If the system absorbs heat or has work done on it (compression), its internal energy increases.
- Energy Out (-q or -w): If the system releases heat or does work on the surroundings (expansion), its internal energy decreases.
| Variable | Process | Sign |
| q | System gains heat (Endothermic) | + |
| q | System loses heat (Exothermic) | – |
| w | Work done on the system (Compression) | + |
| w | Work done by the system (Expansion) | – |
3. Energy is a State Function
A crucial part of the First Law is that Internal Energy (E) is a state function. This means ΔE depends only on the initial and final states of the system, not on the path taken to get there.
However, heat (q) and work (w) are not state functions. You can change the internal energy of a gas by heating it, by compressing it, or a combination of both. While the individual amounts of q and w might vary depending on your method, their sum (ΔE) will always be the same for a specific change in state.
4. Real-World Example: A Car Engine
When fuel burns in a car’s cylinder:
- Heat (q): The combustion reaction releases a massive amount of heat.
- Work (w): That heat causes gas to expand rapidly, pushing the piston down (the system does work on the surroundings).
- Result: The internal energy of the chemical “system” decreases as it is converted into the thermal energy of the exhaust and the mechanical energy (work) that moves the car.
5. Perpetual Motion (Why it’s impossible)
The First Law is the reason “perpetual motion machines of the first kind” cannot exist. You cannot get more work out of a machine than the energy you put into it. If a machine does work, its internal energy must decrease unless it is replenished by heat or more work.
What is ΔE and ΔH and how do they contribute to the heats of reaction?
In thermochemistry, ΔE and ΔH both measure energy changes, but they look at the system through different lenses. Understanding the distinction between them is the key to mastering how heats of reaction are calculated and applied.
1. Internal Energy Change (ΔE)
ΔE represents the total change in the system’s internal energy. It accounts for every way energy can enter or leave: both heat (q) and work (w).
ΔE = q + w
When a reaction occurs in a sealed, rigid container (like a bomb calorimeter), the volume cannot change. This means the system can’t do any expansion work (w = 0). In this specific case, all the energy released or absorbed by the reaction is heat:
ΔE = qv (heat at constant volume)
2. Enthalpy Change (ΔH)
ΔH is a specialized version of energy change. Most chemistry happens in open beakers or in our atmosphere, where the pressure remains constant but the volume can change (like a gas expanding).
Enthalpy is defined as H = E + PV. Therefore, the change in enthalpy is:
ΔH = ΔE + PΔV
When we substitute the First Law (ΔE = q + w) and the definition of expansion work (w = -ΔV), we find a beautiful simplification:
ΔH = (qp + w) – w = qp
ΔH is simply the heat exchanged at constant pressure (qp).
3. How They Contribute to Heats of Reaction
The “Heat of Reaction” usually refers to ΔH because most lab experiments and biological processes occur at constant atmospheric pressure.
| Term | What it measures | Where it’s used |
| ΔE | Total energy change (Heat + Work) | Rigid containers, combustion science, and physics. |
| ΔH | Heat flow only (at constant pressure) | General chemistry, biology, and weather science. |
The “Work” Difference
The difference between ΔE and ΔH is essentially the energy “spent” on moving the surroundings.
- If a reaction produces a lot of gas (like an explosion), the system must “push” the atmosphere out of the way to make room.
- Some of the reaction’s total energy (ΔE) is used to do that work.
- The remaining energy that we actually feel as a temperature change is the enthalpy (ΔH).
4. Why the Difference is Usually Small
For reactions involving only solids and liquids, the volume change ($\Delta V$) is almost zero. In these cases, ΔE and ΔH are virtually identical. The difference only becomes significant when gases are produced or consumed, as gases occupy much more volume than solids or liquids.
ΔH = ΔE + ΔngasRT
where Δngas is the change in the number of moles of gas in the reaction.
What is the indirect determination of ΔH and what is Hess’s Law?
Sometimes, it is physically impossible or extremely dangerous to measure the enthalpy of a reaction directly in a calorimeter. For these situations, we use an indirect determination method known as Hess’s Law.
1. What is Hess’s Law?
Hess’s Law of Constant Heat Summation states that if a reaction is carried out in a series of steps, the total enthalpy change (ΔH) for the overall reaction is equal to the sum of the enthalpy changes for the individual steps.
This works because enthalpy is a state function. It doesn’t matter if you go from Reactants to Products in one giant leap or three small steps; the total change in “altitude” (energy) is the same.
2. The Rules of the “Hess’s Law Game”
To calculate an unknown ΔH, you can manipulate known chemical equations like algebraic equations. However, you must follow these two rules:
- Rule of Reversal: If you reverse a chemical reaction (flip the products and reactants), you must change the sign of ΔH.
- Example: If A → B has ΔH = +10 kJ, then B → A has ΔH = -10 kJ.
- Rule of Multiplication: If you multiply the coefficients of a reaction by a factor (n), you must multiply ΔH by that same factor.
- Example: If A → B has ΔH = +10 kJ, then 2A → 2B has ΔH = +20 kJ.
3. Why Use Indirect Determination?
There are three main reasons why we can’t always just “measure it”:
- The reaction is too slow: Some reactions (like carbon turning into diamond) take millions of years.
- Side reactions: Sometimes, when you mix A and B, they create C, D, and E all at once, making it impossible to isolate the heat of just one specific product.
- Safety: The reaction might be too explosive or involve highly toxic intermediates that shouldn’t be handled in a standard calorimeter.
4. Standard Enthalpies of Formation (ΔHf°)
The most common way to use Hess’s Law indirectly is by using a “cheat sheet” of Standard Enthalpies of Formation. This is the energy change when 1 mole of a substance is formed from its elements in their most stable form (like O2 gas or C graphite).
The formula is:
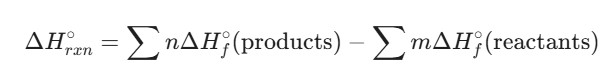
Essentially, you are “deconstructing” the reactants back into their elements (zero energy state) and then “reconstructing” them into products.
5. Practical Example: Carbon Monoxide
Burning graphite to make carbon dioxide (CO2) is easy to measure. But burning graphite to make pure carbon monoxide (CO) is hard because some CO2 always forms.
- By knowing the ΔH for C + O2 → CO2
- And knowing the ΔH for CO + (1/2)O2 → CO2
- We can mathematically subtract them to find the “invisible” heat of reaction for making CO directly.
What are standard enthalpies of formation?
To understand Standard Enthalpies of Formation (ΔHf°), think of them as the “sea level” or the “zero point” of chemical energy. Since it is impossible to measure the absolute enthalpy of a substance, chemists instead measure the change in energy when a substance is built from its most basic, natural parts.
1. The Formal Definition
The Standard Enthalpy of Formation is the change in enthalpy that occurs when exactly 1 mole of a compound is formed from its constituent elements in their standard states.
- Standard State: The physical state (solid, liquid, or gas) a substance naturally exists in at 1 atm of pressure and a specific temperature (usually 25°C or 298.15 K).
- The “Zero” Rule: By definition, the ΔHf° of an element in its most stable form is zero. For example, O2(g), C(graphite), and Na(s) all have a ΔHf° = 0.
2. The Formation Equation
When writing a formation equation, you must ensure that only one mole of the product is created. This often requires using fractions for the reactants, which is perfectly acceptable in thermochemistry.
Example: The formation of liquid water (H2O)
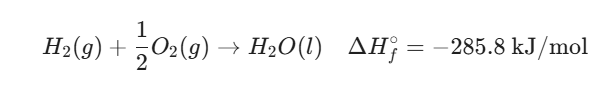
In this case, 285.8 kJ of heat is released when one mole of water is formed from hydrogen and oxygen gas.
3. Using ΔHf° to Find the Heat of Reaction
The most powerful use of these values is calculating the enthalpy change for any reaction without ever stepping into a lab. You simply subtract the total energy of the “starting materials” from the “finished products.”
The formula is:

- n and m: The stoichiometric coefficients from the balanced chemical equation.
- Σ: The sum of all parts.
4. Why is this useful?

Imagine you want to calculate the energy released by burning propane (C3H8). Instead of measuring it, you look up the “cost” to build propane, CO2, and H2O in a reference table:
- “Deconstruct” the propane into carbon and hydrogen (this costs the reverse of its formation energy).
- “Construct” the CO2 and H2O from those same elements.
- The difference between those two steps is your Heat of Combustion.
5. Stability and ΔHf°
The value of ΔHf° tells you a lot about the stability of a compound:
- Large Negative Value: The compound is very stable compared to its elements (energy was released to form it).
- Positive Value: The compound is relatively unstable or “high energy” (energy had to be forced into the bonds to create it).
What are fuels as a source of energy?
In thermochemistry, fuels are substances that release significant amounts of energy—usually as heat—during a chemical reaction, most commonly combustion.
When we talk about fuels “containing” energy, we are specifically referring to the chemical potential energy stored in the arrangement of their atoms and bonds.
1. How Fuels Release Energy
Almost all common fuels (wood, coal, gasoline, natural gas) are composed of hydrocarbons—molecules made of carbon and hydrogen.
The energy release happens during a combustion reaction where the fuel reacts with oxygen ($O_2$):
- Breaking Bonds: It takes energy to break the strong C-H and C-C bonds in the fuel.
- Forming Bonds: A massive amount of energy is released when the atoms reform into very stable C=O bonds (in CO2) and O-H bonds (in H2O).
Because the energy released from forming the new bonds is much greater than the energy required to break the old ones, the overall process is highly exothermic ($-\Delta H$).
2. Common Types of Fuels
Fuels are generally categorized by their origin and state of matter:
- Fossil Fuels: Created from the remains of ancient plants and animals over millions of years.
- Coal: High carbon content; releases the most CO2 per unit of energy.
- Petroleum (Oil): Refined into gasoline, diesel, and jet fuel.
- Natural Gas (Methane, CH4): Burns more “cleanly” than coal or oil, producing less CO2 and fewer pollutants.
- Biofuels: Derived from contemporary biological processes (like ethanol from corn or biodiesel from vegetable oils).
- Hydrogen (H2): A high-energy fuel that produces only water vapor (H2O) as a byproduct. However, it is difficult to store and currently expensive to produce.
3. Fuel Value (Energy Density)
The fuel value is the amount of energy released by the combustion of a specific mass of fuel (usually expressed in kJ/g).
| Fuel Source | Energy Density (approx. kJ/g) | Main Component |
| Wood | 18 kJ/g | Cellulose |
| Coal (Anthracite) | 30 kJ/g | Carbon |
| Gasoline | 45 kJ/g | Octane (C8H18) |
| Natural Gas | 50 kJ/g | Methane (CH4) |
| Hydrogen | 142 kJ/g | H2 |
4. The Efficiency Problem

Not all the energy released by a fuel is turned into useful work. According to the laws of thermodynamics, some energy is always “lost” as waste heat to the surroundings.
- A typical internal combustion engine might only be 20–30% efficient, meaning 70% of the fuel’s energy is wasted as heat through the exhaust and radiator.
- Power plants are often more efficient (around 40–60%) but still lose significant energy during the conversion from heat to electricity.
5. Environmental Impact
From a thermochemical perspective, the “cost” of using hydrocarbon fuels is the production of Greenhouse Gases.
- CO2 Production: Since carbon is the backbone of these fuels, CO2 is an inevitable product of their combustion.
- Heat Pollution: Large-scale energy release can influence local microclimates (like the “urban heat island” effect).
How does AI relate to thermochemistry?
The intersection of Artificial Intelligence and thermochemistry is a rapidly expanding field, primarily focused on overcoming the “computational bottleneck” of traditional physical chemistry. While classical thermochemistry relies on experiments (calorimetry) or intense calculus-based simulations, AI accelerates these processes by predicting molecular behavior.
Here is how AI is currently transforming the domain:
1. Neural Network Potentials (NNPs)
Traditionally, calculating the Internal Energy (ΔE) of a complex molecule requires solving Schrödinger’s equation—a task that is mathematically exhausting even for supercomputers.
AI models, specifically Graph Neural Networks (GNNs), are trained on databases of known molecular structures. They can “predict” the potential energy surface of a new molecule in milliseconds rather than days. This allows researchers to calculate enthalpies of formation for thousands of hypothetical compounds at a fraction of the cost.
2. High-Throughput Material Discovery
AI is used to scan “chemical space” to find new fuels or materials for thermal energy storage.
- Catalysis: AI models predict which surface structures will lower the Activation Energy (Ea) of a reaction most effectively.
- Battery Tech: Researchers use machine learning to model the heat generated during charge/discharge cycles (ΔHcycling), helping to design safer, cooler batteries for electric vehicles.
3. Predictive Calorimetry
Instead of performing a physical experiment for every new substance, AI can estimate Specific Heat Capacities (c) and Standard Enthalpies (ΔHf°) based on molecular descriptors (like bond types, atom counts, and spatial geometry).
- These models use Regression Analysis to find patterns in vast tables of thermochemical data.
- They are particularly useful for volatile or rare substances where physical calorimetry is dangerous or too expensive.
4. Atmospheric and Meteorological Modeling
Since thermochemistry governs the phase changes of water and the movement of energy in the air, AI is being integrated into weather prediction:
- Latent Heat Exchange: AI models better simulate how energy is released during condensation in clouds, which is a key driver for storm intensity.
- Combustion Emissions: AI tracks the thermochemical breakdown of pollutants in the atmosphere, predicting how NOx and SOx gases will react under different thermal conditions.
5. Optimizing Industrial Thermochemical Processes
In large-scale chemical plants (like those producing ammonia or refining oil), AI “digital twins” monitor real-time heat flow.

- The Goal: Maximize the First Law efficiency (ΔE = q + w).
- The Method: AI adjusts pressures and temperatures in real-time to ensure the reaction stays in the most energy-efficient “sweet spot,” reducing waste heat and fuel consumption.
Summary: From Calculus to Connections
In the past, a thermochemist’s best friend was a massive table of ΔHf° values and a calculator. Today, it is a Generative Model that can simulate how energy flows through a system before the chemicals ever touch a beaker.
