Thermochemistry is the study of energy and heat associated with chemical reactions and physical transformations. By understanding how energy moves between a system and its surroundings, we can predict reaction behavior, calculate fuel efficiency, and even model atmospheric weather patterns.
1. The Language of Energy: Key Terminology
To navigate thermochemistry, we must first define our “map” of energy transfer:
- The System vs. Surroundings: The “system” is the specific chemical reaction being studied, while the “surroundings” include everything else (the beaker, the air, the scientist).
- Enthalpy (ΔH): The total heat content of a system at constant pressure.
- Exothermic (-ΔH): Heat exits the system; surroundings get hotter (e.g., combustion).
- Endothermic (+ΔH): Heat enters the system; surroundings get colder (e.g., melting ice).
- State Functions: Properties like Enthalpy and Internal Energy (E) that depend only on the current state, not the path taken to get there.
2. Heat and Work: The Drivers of Change
Internal Energy (ΔE) changes through two primary mechanisms: Heat (q) and Work (w).
- Heat (q): Energy transfer due to temperature differences. It is calculated using specific heat capacity: q = m * c * ΔT.
- Work (w): Energy transfer through organized motion, typically Pressure-Volume (PV) work in chemistry (w = -PΔV).
- The First Law of Thermodynamics: Energy cannot be created or destroyed. The change in a system’s internal energy is the sum of heat and work: ΔE = q + w.
3. Measuring and Calculating Enthalpy
Since we cannot measure total enthalpy directly, we use two primary methods to find the Heat of Reaction:
- Calorimetry: An experimental technique where we measure the temperature change of a surrounding medium (usually water) to calculate the energy released or absorbed by a reaction.
- Hess’s Law: A mathematical approach stating that the total enthalpy change is the same regardless of the number of steps in a reaction. This allows us to use Standard Enthalpies of Formation (ΔHf°)—the “sea level” of chemical energy—to calculate the heat of any reaction:
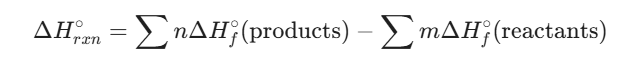
4. Fuels and Energy Density
Fuels are substances that store high chemical potential energy in their bonds.
- Hydrocarbons: Most fuels (gasoline, methane, coal) release energy when their C-H bonds are broken and more stable C=O and O-H bonds are formed.
- Fuel Value: The energy released per gram of fuel. Hydrogen holds the highest energy density (142 kJ/g), while coal and wood provide significantly less.
5. The Future: AI in Thermochemistry
Artificial Intelligence is transforming how we calculate energy states by overcoming the “computational bottleneck” of manual calculus and complex simulations.
- Neural Network Potentials (NNPs): AI can predict the potential energy of complex molecules in milliseconds.
- Material Discovery: AI identifies new catalysts and battery materials by modeling heat generation and activation energy.
- Meteorological Impact: AI enhances weather forecasting by more accurately simulating latent heat exchange during water phase changes in the atmosphere.
SEO Summary Keywords
- Primary Keywords: Thermochemistry, Enthalpy (ΔH), First Law of Thermodynamics, Calorimetry, Hess’s Law.
- Secondary Keywords: Exothermic vs Endothermic, Specific Heat Capacity, Standard Enthalpy of Formation, Internal Energy, PV Work, AI in Chemistry.
- Search Intent: Educational resource for chemistry students, professionals in geomatics/meteorology, and energy science enthusiasts.
