How can chemistry be considered the blueprint of matter?
Earth and Atmospheric Sciences
Filter – PhET Simulations – Chemistry
General Chemistry AI tutor | Course problem & homework solver
Gases are one of the four fundamental states of matter (along side solids, liquids, and plasma). Unlike solids or liquids, gas particles have enough kinetic energy to completely overcome the attractive forces that would otherwise hold them together.
Here is a breakdown of how gases behave and the “rules” they follow:
1. The Kinetic Molecular Theory
To understand what a gas is, we look at the Kinetic Molecular Theory, which describes gas particles as being in constant, random motion.
- Empty Space: Most of a gas is actually empty space. The particles themselves take up very little volume compared to the total space they occupy.
- Constant Motion: Particles move in straight lines until they collide with each other or the walls of a container.
- Elastic Collisions: When gas particles hit something, they don’t lose energy; they just bounce off and keep moving.
2. Distinct Physical Characteristics
Because of their high energy and weak attractions, gases have unique properties:

- Expansion: A gas will expand to fill the entire shape and volume of whatever container it is in.
- Compressibility: Because there is so much space between particles, you can easily “squeeze” a gas into a smaller volume (like filling a scuba tank or a tire).
- Low Density: Gases have much lower densities than solids or liquids because the particles are so spread out.
- Diffusion: Gases mix rapidly with one another. This is why you can smell coffee brewing from across a room almost instantly.
3. The Variables of Gas (The Ideal Gas Law)
In chemistry and physics, the behavior of a gas is defined by the relationship between four variables: Pressure (P), Volume (V), Temperature (T), and the Amount of gas (n).
These are mathematically linked by the Ideal Gas Law:
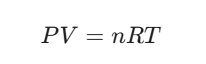
- Where R is the universal gas constant.
- This equation shows that if you heat a gas (increase T) in a fixed container, the pressure (P) will rise because the particles are hitting the walls harder and more often.
4. Real vs. Ideal Gases

In a laboratory setting, we often talk about “Ideal Gases”—imaginary gases that follow the rules perfectly. However, Real Gases (like the Nitrogen and Oxygen in our atmosphere) behave slightly differently at extremely high pressures or very low temperatures because the particles eventually get close enough to start attracting one another.
Common Gases in Our Environment
| Gas | Composition | Common Role |
| Nitrogen (N2) | ~78% of air | Provides atmospheric pressure; relatively inert. |
| Oxygen (O2) | ~21% of air | Essential for respiration and combustion. |
| Argon (Ar) | ~0.93% of air | A noble gas used in specialized lighting and welding. |
| Carbon Dioxide (CO2) | Trace amount | Vital for photosynthesis and a key greenhouse gas. |
What are the properties of gases and what is gas pressure?
Gases are unique because their particles have enough energy to overcome the attractive forces that keep solids and liquids together. This high-energy state results in several distinct physical properties and the phenomenon of pressure.
Core Properties of Gases
Gases are defined by their lack of fixed shape and volume. Because the particles are so far apart and moving so fast, they exhibit the following behaviors:
- Expansion: A gas will expand to fill the entire volume and shape of its container. There is no “surface” to a gas like there is with a liquid.
- Compressibility: Because there is so much empty space between gas atoms or molecules, they can be squeezed into a much smaller volume. This is why we can store large amounts of oxygen or propane in metal tanks.
- Low Density: Compared to solids and liquids, gases have very little mass per unit of volume. For example, liquid water is about 800 times denser than water vapor at sea level.
- Diffusion and Effusion: Gases mix rapidly with one another (diffusion) and can pass through tiny openings (effusion). This is why the scent of a perfume can quickly fill a large room.
What is Gas Pressure?

At the molecular level, gas pressure is the result of countless microscopic collisions.
When gas particles move, they constantly strike the walls of their container. Each individual collision exerts a tiny “push.” When you sum up billions of these collisions occurring every second over a specific area, you get a measurable force.
The Mathematical Definition
In physics and chemistry, pressure (P) is defined as the force (F) applied perpendicular to the surface of an object per unit area (A) over which that force is distributed:
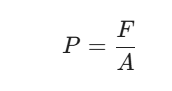
Factors That Change Pressure
Based on the Kinetic Molecular Theory, three main factors influence how much pressure a gas exerts:
- Temperature: If you heat a gas, the particles move faster. Faster particles hit the walls more often and with more force, increasing the pressure.
- Volume: If you shrink the container (decreasing volume) without changing the amount of gas, the particles hit the walls more frequently because they have less distance to travel. This increases pressure.
- Amount of Gas: Adding more particles to a container increases the total number of collisions, which raises the pressure.
Measuring Pressure
Pressure can be measured in several different units depending on the context, such as meteorology, engineering, or chemistry:
| Unit | Common Usage |
| Atmosphere (atm) | Standard reference based on air pressure at sea level. |
| Pascal (Pa) / kPa | The SI unit; common in physics and Canadian weather reports. |
| Millimeters of Mercury (mmHg) | Often used in medicine (blood pressure) and barometers. |
| PSI (Pounds per Square Inch) | Common in mechanical engineering and tire pressure. |
What are the simple gas laws?
The simple gas laws describe how the state of a gas—its Pressure (P), Volume (V), and Temperature (T)—changes when one variable is held constant. These laws were discovered by scientists observing how gases behave in closed systems.
1. Boyle’s Law: Pressure and Volume
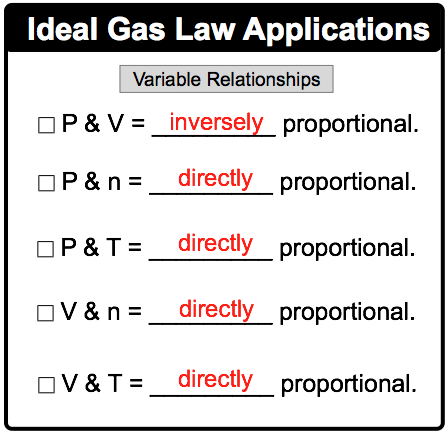
Discovered by Robert Boyle, this law states that at a constant temperature, the pressure of a gas is inversely proportional to its volume.
- The Concept: If you compress a gas into a smaller space, the particles hit the walls more often, increasing the pressure.
- The Formula: P1 * V1 = P2 * V2
2. Charles’s Law: Volume and Temperature
Jacques Charles found that at a constant pressure, the volume of a gas is directly proportional to its absolute temperature (measured in Kelvin).
- The Concept: As you heat a gas, the particles move faster and push outward more strongly. If the container can expand (like a balloon), the volume increases.
- The Formula: V1 / T1 = V2 / T2
3. Gay-Lussac’s Law: Pressure and Temperature
This law states that at a constant volume, the pressure of a gas is directly proportional to its absolute temperature.
- The Concept: If you heat a gas in a rigid container (like a pressure cooker or a metal tank), the particles hit the walls with more force, but the volume cannot change, so the pressure rises.
- The Formula: P1 / T1 = P2 / T2
4. Avogadro’s Law: Volume and Amount
Periodic Table of the Elements, With Symbols – Map Quiz Game – Seterra
Amedeo Avogadro proposed that at a constant temperature and pressure, the volume of a gas is directly proportional to the number of moles (n) of the gas.
- The Concept: If you add more gas particles to a flexible container, the volume must increase to maintain the same pressure.
- The Formula: V1 / n1 = V2 / n2
Summary Table: Gas Law Relationships
| Law | Constant | Relationship | Real-World Example |
| Boyle’s | Temperature | P ↑, V ↓ | Squeezing a balloon. |
| Charles’s | Pressure | T ↑, V ↑ | A basketball going flat in the cold. |
| Gay-Lussac’s | Volume | T ↑, P ↑ | Pressure building in a car tire on a hot day. |
| Avogadro’s | P and T | n ↑, V ↑ | Blowing air into a lung or balloon. |
What is the ideal gas equation?
The Ideal Gas Equation is the mathematical relationship that brings all the simple gas laws together into one single formula. It describes the state of a “perfect” gas by linking pressure, volume, temperature, and the amount of substance present.
The Equation
The formula is written as:
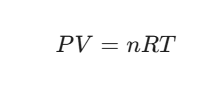
Where:
- P is the Pressure (measured in atmospheres, kilopascals, or mmHg).
- V is the Volume (usually measured in liters).
- n is the Amount of Substance (measured in moles).
- R is the Ideal Gas Constant, which acts as the bridge between the variables.
- T is the Absolute Temperature (must be measured in Kelvin).
What is the Gas Constant (R)?
The value of R depends on the units you use for pressure. It is a proportionality constant that ensures the two sides of the equation balance out. Common values include:
- 0.0821 (L • atm) / (K • mol)
- 8.314 J / (K • mol) (used when pressure is in Pascals and volume is in cubic meters)
Why is it “Ideal”?
In the real world, no gas is truly “ideal.” An ideal gas is a theoretical model based on two main assumptions:

- The gas particles take up zero volume themselves.
- There are no attractive or repulsive forces between the particles.
While most gases (like Nitrogen, Oxygen, or Hydrogen) behave very much like an ideal gas under standard conditions, they begin to deviate from this equation at extremely high pressures (where particles are forced close together) or extremely low temperatures (where particles move slowly enough for intermolecular forces to take over).
Practical Applications
This equation is a fundamental tool for scientists and engineers. It allows you to:
- Calculate Density: By rearranging the formula, you can find the density of a gas at a specific altitude or temperature.
- Determine Molar Mass: If you know the mass of a gas sample and its P, V, and T, you can identify what substance it is.
- Safety Engineering: It helps in calculating the maximum pressure a storage tank can withstand as temperatures fluctuate.
What are applications of the ideal gas equation?
The Ideal Gas Equation (PV = nRT) is a cornerstone of both theoretical science and practical engineering. It serves as a mathematical bridge that allows us to predict how matter will behave under different environmental conditions.

Here are the primary applications where this “blueprint” is put to work:
1. Atmospheric Science and Meteorology
One of the most frequent uses of the ideal gas law is in understanding our atmosphere. It helps scientists calculate the density of air at various altitudes.
- Lifting Force: By comparing the density of a gas inside a balloon (like Helium or heated air) to the density of the surrounding atmosphere calculated via PV=nRT, we can determine the buoyancy or lifting capacity of weather balloons.
- Pressure Gradients: It is used to model how pressure changes with temperature, which is essential for forecasting wind patterns and storm systems.
2. Engineering and Safety
Engineers rely on this equation to design systems that handle compressed gases safely.
- Storage Tanks: When designing tanks for oxygen, propane, or nitrogen, engineers use the equation to predict how much the internal pressure will rise if the tank is sitting in the sun on a hot day.
- Automotive Safety: The rapid inflation of airbags is a practical application. A chemical reaction produces nitrogen gas (n); the equation helps determine exactly how many moles are needed to fill the volume (V) of the bag to the correct pressure (P) in milliseconds.
3. Determining Molar Mass and Density

In a laboratory setting, the ideal gas law is a powerful tool for identifying unknown substances.
- Molar Mass: If you have a sample of an unknown gas, you can measure its mass, pressure, volume, and temperature. By rearranging the equation to n = (PV) / (RT), you can find the number of moles. Dividing the mass by the moles gives you the molar mass, which helps identify the gas.
- Gas Density: The equation can be rearranged to relate density (ρ) directly to pressure and temperature: ρ = (PM) / (RT) (Where M is the molar mass).
4. Aerospace and Aviation
The equation is vital for calculating engine performance and cabin pressurization.
- Combustion Engines: In both car engines and jet turbines, the equation helps calculate the expansion of gases during the combustion stroke, which is what actually generates the mechanical work (power).
- Altimeters: Many aircraft instruments use the relationship between pressure and altitude (tempered by temperature) to provide accurate height readings to pilots.
Summary of Applications
| Field | Specific Use | Variable of Interest |
| Chemistry | Identifying unknown gases | Molar Mass (M) |
| Meteorology | Weather balloon buoyancy | Density (ρ) |
| Engineering | Airbag deployment | Moles (n) and Volume (V) |
| Automotive | Tire pressure monitoring | Pressure (P) vs. Temperature (T) |
What are gases in chemical reactions?
In chemical reactions, gases can act as reactants, products, or even catalysts. Because gas particles are far apart and move at high velocities, reactions involving gases often happen very quickly or require specific conditions like high pressure to force the particles together.
1. Gases as Reactants
When a gas is a reactant, its concentration is typically measured by its partial pressure. According to collision theory, increasing the pressure of a reactant gas increases the frequency of collisions, which speeds up the reaction rate.
- Combustion: One of the most common reactions where oxygen (O2) gas reacts with a fuel (like methane or wood) to produce energy.
- Haber Process: A massive industrial reaction that combines Nitrogen (N2) and Hydrogen (H2) gases to create ammonia for fertilizer. N2(g) + 3H2(g) ⇌ 2NH3 (g)
2. Gases as Products
Many reactions produce a gas as a byproduct. In a laboratory setting, this is often observed as effervescence (bubbling).
- Metal and Acid: When a metal like zinc reacts with hydrochloric acid, it produces Hydrogen gas (H2).
- Decomposition: Heating calcium carbonate (limestone) causes it to break down into solid calcium oxide and Carbon Dioxide gas (CO2).
3. Stoichiometry and Volume
Because of the Ideal Gas Law, we can use the volume of a gas to determine how much of a solid or liquid reactant was used. At Standard Temperature and Pressure (STP), 1 mole of any gas occupies 22.4 Liters.
This allows chemists to calculate the “yield” of a reaction simply by measuring the volume of gas collected in a graduated cylinder or a gas syringe.
4. Gas Evolution Reactions
A “gas evolution reaction” is a specific type of reaction that forms a gas upon mixing two liquids or a solid and a liquid. These are often used in:
- Safety Devices: The decomposition of sodium azide (NaN3) into Nitrogen gas to inflate airbags.
- Leavening: Baking powder reacts with moisture and heat to produce CO2 gas, which makes bread and cakes rise.
Summary Table: Gases in the Lab
| Role | Example Reaction | Visible Sign |
| Reactant | Hydrogenation of oils | Decrease in system pressure |
| Product | Vinegar and baking soda | Rapid bubbling/fizzing |
| Intermediate | Internal combustion | Rapid expansion and work |
What is the kinetic-molecular theory of gases?
The Kinetic-Molecular Theory (KMT) is the theoretical model that explains why gases behave the way they do. While the gas laws (like Boyle’s or Charles’s) describe what happens, KMT explains the underlying motion of particles that causes those observations.
It is based on five core postulates:
1. Constant, Random Motion
Gas particles are in continuous, rapid, and random motion. They move in straight lines until they collide with each other or the walls of their container. This explains why gases expand to fill any space available to them.
2. Negligible Particle Volume
The actual molecules of a gas are so tiny compared to the distances between them that their individual volume is considered to be zero. A gas is mostly empty space. This is why gases are so easily compressed compared to solids.
3. No Intermolecular Forces
KMT assumes there are no forces of attraction or repulsion between gas particles. They don’t “stick” together when they pass each other; they act like perfectly independent entities.
4. Elastic Collisions
When gas particles collide with each other or the container walls, the collisions are perfectly elastic. This means there is no net loss of total kinetic energy. If energy were lost during collisions, the gas particles would eventually slow down, clump together, and the gas would collapse into a liquid.
5. Temperature and Kinetic Energy
The average kinetic energy of gas particles is directly proportional to the absolute temperature (Kelvin) of the gas.
- At higher temperatures, particles move faster and hit harder.
- All gases at the same temperature have the same average kinetic energy, regardless of their identity.

How KMT Explains the Gas Laws
| Observation | KMT Explanation |
| Pressure | Pressure is caused by the force of billions of particles hitting the container walls. |
| Boyle’s Law | Decreasing volume means particles hit the walls more often, increasing pressure. |
| Charles’s Law | Increasing temperature makes particles move faster; they hit the walls harder, pushing them outward and increasing volume. |
| Diffusion | Because particles are in constant motion and have mostly empty space to move through, they mix quickly. |
Real Gas Deviations
It is important to note that KMT describes an Ideal Gas. In the real world, “Real Gases” eventually deviate from these rules—especially at very high pressures (where particle volume matters) or very low temperatures (where attractive forces start to pull particles together).
What are gas properties relating to the kinetic-molecular theory?
The Kinetic-Molecular Theory (KMT) provides the “why” behind the physical properties we observe in gases. By viewing gas as a collection of rapidly moving particles, we can connect microscopic behavior to macroscopic characteristics.
Here are the primary gas properties explained through the lens of KMT:
1. Pressure: The Result of Collisions
In KMT, pressure isn’t just a static force; it is the cumulative result of billions of tiny impacts. Every time a gas particle strikes the wall of a container, it exerts a force.
- The KMT Link: Since particles are in constant, random motion, they are always hitting the walls. If you increase the speed of the particles (temperature) or put more particles in the box, the frequency and force of these collisions increase, raising the pressure.
2. Compressibility: The Vast Empty Space
Gases are highly compressible, unlike solids or liquids.
- The KMT Link: One of the core postulates of KMT is that particle volume is negligible compared to the total volume of the gas. Because a gas is mostly empty space, you can push the particles closer together without them physically pushing back until they are nearly touching.

3. Expansion: Overcoming Attraction
A gas will expand to fill any container, regardless of its size or shape.
- The KMT Link: KMT assumes no intermolecular forces (attraction or repulsion) between particles. Because the particles don’t “stick” to each other and are moving at high velocities, they simply keep traveling in straight lines until they hit a boundary, effectively filling the entire space.
4. Diffusion and Effusion: Path of Least Resistance
- Diffusion is the mixing of gases (like smelling perfume across a room).
- Effusion is gas escaping through a tiny hole (like a balloon deflating over time).
- The KMT Link: Because particles move in random straight lines and the space between them is mostly empty, they can travel long distances and weave past other particles easily. Faster-moving (lighter) particles will diffuse and effuse more quickly than heavier ones.
5. Temperature: The Measure of Motion
We perceive temperature as “heat,” but KMT redefines it as mechanical energy.
- The KMT Link: The average kinetic energy of the particles is directly proportional to the Kelvin temperature. This is why a gas at 600 K has particles moving significantly faster than the same gas at 300 K. It also explains why different gases at the same temperature exert the same average kinetic energy, regardless of their mass.
Summary: From Theory to Property
| Gas Property | Related KMT Postulate |
| Pressure | Constant, random motion and collisions. |
| Compressibility | Negligible particle volume (mostly empty space). |
| Expansion | No intermolecular forces of attraction. |
| Low Density | Particles are far apart with little mass per volume. |
| Temperature | Proportional to average kinetic energy (KEavg). |
What are nonideal (real) gases?
While the Ideal Gas Law provides a great “blueprint” for how gases should behave, Real Gases are those that deviate from these theoretical rules under specific conditions. In the real world, the two “rules” we assume for ideal gases—that particles have no volume and no attraction to each other—simply aren’t true.
Why Real Gases Differ from Ideal Gases
The Kinetic-Molecular Theory assumes that gas particles are “point masses” (taking up no space) and that they don’t care about their neighbors. However, real atoms and molecules have physical characteristics that matter when they get crowded.
1. Particles Have Volume
In an ideal gas, we assume the volume of the particles themselves is zero. In a real gas, as you increase the pressure and force the gas into a smaller space, the volume of the actual molecules becomes a significant portion of the total space. This means the available volume for the gas to move in is actually less than the container’s volume.
2. Intermolecular Forces Exist
Ideal gases are assumed to have no attraction or repulsion. In reality, all molecules have some level of attraction (like London dispersion forces or dipole-dipole interactions). When particles move slowly enough, they “feel” each other’s presence and stick together momentarily, which reduces the force with which they hit the container walls.
When Do Gases Become Non-Ideal?
Gases behave most like an ideal gas at High Temperature and Low Pressure. They become “non-ideal” or “real” under the opposite conditions:
- High Pressure: When you squeeze a gas, the particles are forced so close together that their own physical size (volume) can no longer be ignored.
- Low Temperature: When a gas is cooled, the particles slow down. They no longer have enough kinetic energy to completely overcome their mutual attraction, causing them to “clump” and eventually condense into a liquid.
The van der Waals Equation
To account for these real-world behaviors, scientists use the van der Waals equation. It modifies the Ideal Gas Law (PV=nRT) by adding two constants, a and b, which are unique to every specific gas:
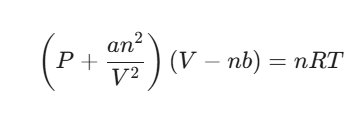
- The a constant: Corrects for the intermolecular attractions that reduce the observed pressure.
- The b constant: Corrects for the finite volume of the gas molecules.
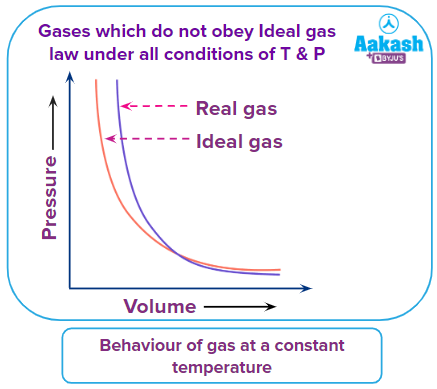
Summary: Ideal vs. Real
| Feature | Ideal Gas | Real Gas |
| Particle Volume | Assumed to be zero. | Small but significant at high pressure. |
| Attraction | No forces between particles. | Particles attract each other at low temperatures. |
| Collisions | Perfectly elastic. | Can be slightly inelastic due to attraction. |
| Equation | PV = nRT | (P + an2 / V2)(V – nb) = nRT |
