How can chemistry be considered the blueprint of matter?
Earth and Atmospheric Sciences
Atomic theory is the scientific framework that describes the nature of matter, stating that everything around us is composed of discrete units called atoms. Rather than matter being infinitely divisible, atomic theory shows us that there is a fundamental “building block” to the universe.
Over centuries, our understanding has shifted from seeing atoms as solid “billiard balls” to complex systems of probability and energy.
How Atomic Theory Describes the Atom
Modern atomic theory defines the atom not just by what it is, but by how its subatomic particles interact.
1. The Nucleus (The Core)
At the center of every atom lies a dense nucleus. It contains two main types of nucleons:
- Protons: Positively charged particles. The number of protons determines the atomic number and, essentially, the identity of the element (e.g., 6 protons always make Carbon).+1
- Neutrons: Particles with no charge (neutral). They act as a sort of “nuclear glue,” helping to stabilize the protons within the nucleus.
2. The Electron Cloud
Instead of orbiting the nucleus like planets around a sun (an older model known as the Bohr model), modern theory describes electrons existing in orbitals.
- Electrons: Negatively charged particles that are significantly smaller than protons and neutrons.
- Probability: We don’t know exactly where an electron is at any given moment; instead, we describe “clouds” or regions of space where they are most likely to be found.
The Evolution of the Model
The theory has evolved as our tools for peering into the microscopic world improved:
| Scientist | Model Name | Key Concept |
| John Dalton | Billiard Ball | Atoms are solid, indivisible spheres. |
| J.J. Thomson | Plum Pudding | Discovered electrons embedded in a positive “soup.” |
| Ernest Rutherford | Nuclear Model | Discovered the dense, positive nucleus via the Gold Foil experiment. |
| Niels Bohr | Planetary Model | Electrons move in fixed orbits or energy levels. |
| Schrödinger/Heisenberg | Quantum Model | Electrons exist in “clouds” of probability. |
Key Principles
- Mass Distribution: Nearly all of an atom’s mass is in the nucleus, but nearly all of its volume is empty space occupied by the electron cloud.
- Electrostatic Attraction: The atom is held together by the electromagnetic force—the negative electrons are attracted to the positive protons in the nucleus.
- Conservation: In standard chemical reactions, atoms are rearranged, but they are never created or destroyed.
How did early chemical discoveries lead to Atomic Theory?
The transition from ancient philosophy to modern science wasn’t an overnight “eureka” moment. It was driven by 18th-century chemists who stopped just observing reactions and started measuring them.
Before we knew what an atom looked like, we discovered how they behaved through three foundational “laws.”
1. The Law of Conservation of Mass
In the late 1700s, Antoine Lavoisier (the “Father of Modern Chemistry”) proved that in a closed system, matter is neither created nor destroyed.
- The Discovery: By carefully weighing reactants and products, he found that the mass at the end of a reaction was identical to the mass at the start.
- The Atomic Link: This suggested that matter is made of “stuff” that can be rearranged, but doesn’t just vanish into thin air.
2. The Law of Definite Proportions
Joseph Proust observed that a specific chemical compound always contains the exact same proportion of elements by mass, regardless of where it came from or how much you have.
- Example: Pure water is always roughly 11% hydrogen and 89% oxygen by mass.
- The Atomic Link: If substances weren’t made of specific, individual units (atoms), you would expect the ratios to be random or messy.
3. The Law of Multiple Proportions
John Dalton took these ideas further. He noticed that if two elements form more than one compound (like Carbon Monoxide and Carbon Dioxide), the masses of one element that combine with a fixed mass of the other are in ratios of small whole numbers.+1
- The Logic: You can have $CO$ or $CO_2$, but you never find $CO_{1.5}$.
- The Atomic Link: This was the “smoking gun.” It implied that elements must be added in discrete, indivisible packets—atoms.
[Image showing the Law of Multiple Proportions with Carbon Monoxide and Carbon Dioxide molecules]
Dalton’s Atomic Theory (1803)
Using these three laws, John Dalton formulated the first scientific atomic theory. His main postulates were:
- All matter is composed of atoms: Tiny, indivisible particles.
- Identity: All atoms of a specific element are identical in mass and properties (we later found out about isotopes, but he was close!).
- Compounds: Compounds are combinations of two or more different types of atoms.
- Rearrangement: A chemical reaction is a rearrangement of atoms.
What changed later?
While Dalton was a genius for his time, he thought atoms were solid, like tiny marbles. It took the discovery of the electron about 100 years later to prove that atoms actually had an internal structure.
What are electrons and what led to other discoveries in atomic physics?
Think of electrons as the “social butterflies” of the atom. While the nucleus stays tucked away in the center, electrons inhabit the vast outer regions, determining how an atom bonds, reacts, and conducts electricity.
What are Electrons?
An electron is a subatomic particle with a negative elementary charge. In the standard model of physics, they are considered elementary particles, meaning they aren’t made of smaller pieces—they are as fundamental as it gets.+1
- Mass: They are incredibly light. A proton is about $1,836$ times more massive than an electron.
- Behavior: They don’t move in neat circles. Instead, they exist in “clouds” of probability defined by quantum mechanics.
- Role: They are responsible for chemical bonding. When atoms share or swap electrons, they form molecules.
The Discovery: The Cathode Ray Experiment
In 1897, J.J. Thomson used a vacuum tube (a cathode ray tube) to show that “rays” emitted from a metal plate were actually streams of particles.
- He applied a magnetic/electric field to the ray.
- The ray bent toward the positive plate.
- The Conclusion: Since opposites attract, the ray must be made of negatively charged particles. He called them “corpuscles,” but we know them as electrons.
How One Discovery Led to the Next
The “Electron” was the first crack in the idea that atoms were indivisible. This triggered a domino effect of discoveries:
1. The Probing of the Nucleus (1911)
If atoms have negative electrons but are overall neutral, there must be a positive charge somewhere. Ernest Rutherford shot alpha particles at thin gold foil.
- The Surprise: Most passed through, but some bounced straight back.
- The Discovery: This proved the atom is mostly empty space with a tiny, dense, positive nucleus at the center.
2. The Neutron “Missing Link” (1932)
Scientists noticed that the atomic number (protons) didn’t account for the total mass of the atom. It was too heavy. James Chadwick discovered the neutron—a particle with mass similar to a proton but with no charge. This explained isotopes (atoms of the same element with different masses).
3. Quantum Mechanics (The 1920s-Present)
As scientists tried to figure out why electrons didn’t just spiral into the nucleus and crash, they realized classical physics didn’t apply.
- Niels Bohr proposed electrons live in specific energy levels (shells).
- Werner Heisenberg and Erwin Schrödinger later proved that electrons behave like both particles and waves, leading to the Cloud Model we use today.
Summary of Subatomic Particles
| Particle | Charge | Location | Discovery Era |
| Electron | Negative (-1) | Orbitals/Cloud | 1897 (Thomson) |
| Proton | Positive (+1) | Nucleus | 1917 (Rutherford) |
| Neutron | Neutral (0) | Nucleus | 1932 (Chadwick) |
What is the nuclear atom?

The nuclear atom is the model of the atom that we recognize today: a tiny, incredibly dense, positively charged nucleus at the center, surrounded by a vast region of empty space where electrons reside.
This concept was a massive departure from previous theories that imagined the atom as a solid, uniform “blob.”
The Breakthrough: Rutherford’s Gold Foil Experiment
In 1911, physicist Ernest Rutherford performed an experiment that changed chemistry forever. He fired alpha particles (heavy, positively charged particles) at a super-thin sheet of gold foil.+1
- The Expectation: Based on the “Plum Pudding” model, the particles should have passed straight through with minimal deflection.
- The Reality: Most passed through, but a small fraction bounced straight back or deflected at sharp angles.
- The Conclusion: Rutherford famously remarked it was as if you fired a 15-inch shell at a piece of tissue paper and it came back and hit you. He concluded that the atom’s positive charge and most of its mass must be concentrated in a tiny central core.
Anatomy of the Nuclear Atom
The nuclear model describes an atom with two distinct regions:
1. The Nucleus (The “Weight”)
- Size: If an atom were the size of a football stadium, the nucleus would be the size of a marble sitting on the 50-yard line.
- Mass: Despite its tiny size, the nucleus contains 99.9% of the atom’s mass.
- Composition: It holds the protons (positive) and neutrons (neutral).
2. The Extranuclear Region (The “Volume”)
- Composition: This is the space outside the nucleus where electrons are found.
- State: It is almost entirely empty space.
- Balance: The negative charge of the electrons in this region perfectly balances the positive charge of the nucleus, making the atom electrically neutral overall.
Why is the Nuclear Atom Important?
Before this model, we didn’t understand how atoms could be stable or why they interacted the way they did. The nuclear model led to several key scientific realizations:
- Atomic Number: We realized that the number of protons in the nucleus defines what the element is.
- The Strong Force: It raised a new question: if protons are all positive and repel each other, what keeps the nucleus together? (This led to the discovery of the Strong Nuclear Force).
- Isotopes: It explained why atoms of the same element could have different weights (different numbers of neutrons in the nucleus).
Comparison: Old vs. New
| Feature | Plum Pudding Model (Pre-1911) | Nuclear Model (Post-1911) |
| Positive Charge | Spread out like “jelly” | Concentrated in the center |
| Mass | Evenly distributed | Concentrated in the nucleus |
| Space | Solid/Filled | Mostly empty space |
What are chemical elements?

A chemical element is a pure substance that consists entirely of one type of atom. They are the primary constituents of matter and cannot be broken down into simpler substances by ordinary chemical means.
If you think of the universe as a giant LEGO set, the elements are the different types of bricks—some are 2×4, some are thin plates, and some are specialized pieces. While you can build complex structures (molecules) with them, the bricks themselves remain what they are.
The Identity of an Element
What makes one element different from another is the number of protons in the nucleus of its atoms. This is known as the Atomic Number (Z).
- Hydrogen (Z=1): Has 1 proton. It is the lightest and most abundant element.
- Carbon (Z=6): Has 6 protons. It is the basis for all known life.
- Gold (Z=79): Has 79 protons. It is a dense, yellow metal.
If you add or remove a proton from an atom, you aren’t just changing its charge; you are literally changing it into a different element.
How Elements are Organized: The Periodic Table
Scientists organize the 118 known elements into the Periodic Table. This table isn’t just a list; it’s a map that groups elements with similar chemical behaviors together.
Key Divisions:
- Metals: Usually shiny, conductive, and malleable (e.g., Iron, Copper, Gold). They make up the majority of the table.
- Nonmetals: Often gases or brittle solids that don’t conduct heat or electricity well (e.g., Oxygen, Nitrogen, Sulfur).
- Metalloids: Elements that have properties of both metals and nonmetals (e.g., Silicon, Arsenic).
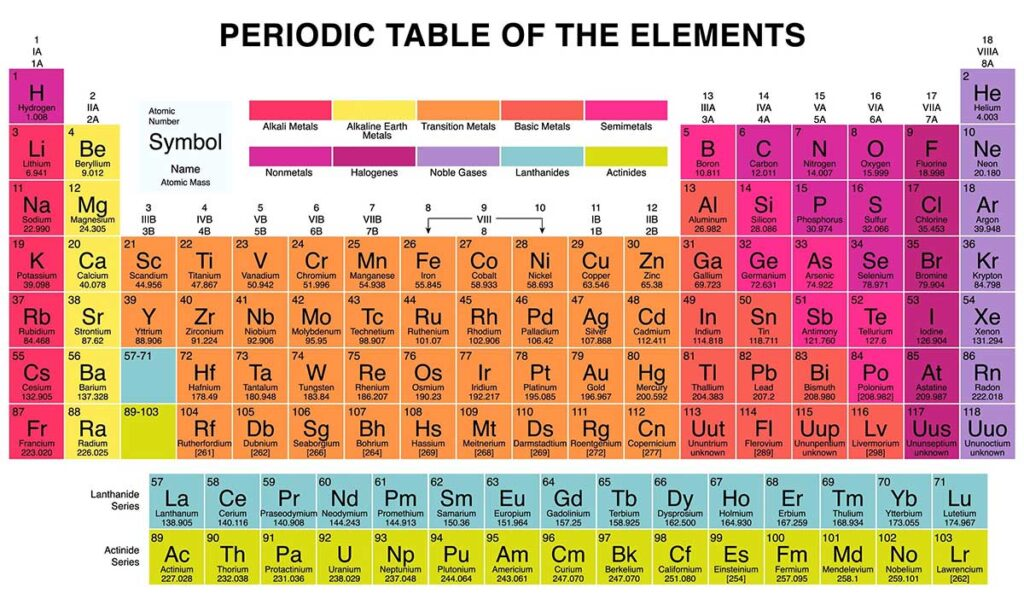
Elements vs. Compounds vs. Mixtures
It is easy to confuse these terms, but they describe how elements interact with each other:
| Type | Definition | Example |
| Element | Only one type of atom. | Pure Gold ($Au$) |
| Compound | Two or more different elements chemically bonded. | Water ($H_2O$) |
| Mixture | Elements or compounds in the same space but NOT bonded. | Air ($N_2, O_2, Ar, CO_2$) |
Where do Elements come from?
- The Big Bang: Produced mostly Hydrogen and Helium.
- Stellar Fusion: Stars “cook” lighter elements into heavier ones, like Carbon and Oxygen.
- Supernovas: The explosion of massive stars creates the heaviest natural elements, like Uranium.
- Synthetic Elements: Elements with atomic numbers 95 through 118 are man-made in particle accelerators and usually exist for only fractions of a second.
What are atomic masses?
To understand atomic mass, it is helpful to think of it as the “weight” of a single atom. Since atoms are far too small to measure on a standard scale, scientists use a specialized system to track how heavy they are relative to one another.
1. Defining Atomic Mass
The atomic mass of an element is the total mass of a single atom. This mass is almost entirely concentrated in the nucleus and is composed of two main particles:
- Protons: Positively charged particles.
- Neutrons: Neutrally charged particles.
Electrons are technically part of the atom, but they are so light (about $1/1800$ the mass of a proton) that their weight is usually ignored in basic calculations.
2. Units of Measurement: The AMU
Because grams are too large for subatomic particles, we use the atomic mass unit (amu).
- 1 amu is defined as exactly 1/12 the mass of a single Carbon-12 atom.
- In this system, a proton and a neutron each have a mass of approximately 1 amu.
3. Relative Atomic Mass (Atomic Weight)
When you look at a Periodic Table, you will notice that the atomic mass is rarely a whole number. For example, Carbon is listed as 12.011, not 12.000. This is because of isotopes.
Most elements exist in nature as a mixture of different “versions” (isotopes) that have the same number of protons but different numbers of neutrons. The number you see on the Periodic Table is a weighted average of all those versions based on how common they are on Earth.
| Particle | Mass (amu) | Location |
| Proton | ~1.007 | Nucleus |
| Neutron | ~1.008 | Nucleus |
| Electron | ~0.0005 | Electron Cloud |
4. Why It Matters
Atomic mass is the bridge between the microscopic world of atoms and the macroscopic world we can actually measure in a lab. It allows chemists to calculate exactly how much of a substance (in grams) is needed to have a specific number of atoms for a chemical reaction, a process known as stoichiometry.
What is the Periodic Table and how does it work?
The Periodic Table is more than just a list of elements; it is a masterpiece of scientific organization. It arranges all known chemical elements in a way that reveals patterns in their properties, allowing scientists to predict how an element will behave before they even touch it.+1

1. The Basic Layout: Periods and Groups
The table is organized by Atomic Number (the number of protons), starting from Hydrogen (1) and moving left-to-right, top-to-bottom.
Periods (Rows)
- What they represent: A horizontal row is called a period.
- The Science: Elements in the same period have the same number of electron shells (energy levels). As you move across a period, the atoms get smaller and more “tightly packed” because the increasing number of protons pulls the electrons closer.
Groups (Columns)
- What they represent: A vertical column is called a group or family.
- The Science: This is the most important part of the table. Elements in a group have the same number of valence electrons (outermost electrons). Because valence electrons dictate how an atom bonds, elements in a group share very similar chemical personalities.+1
2. Regions of the Table
The table is generally divided into three main categories based on physical and chemical properties:
| Category | Location | Properties |
| Metals | Left and Center | Shiny, conduct heat/electricity, malleable. |
| Nonmetals | Upper Right | Poor conductors, often gases or brittle solids. |
| Metalloids | The “Staircase” | Properties of both (e.g., Silicon). |
3. Notable “Families”
Some groups are so distinct they have their own names:
- Group 1: Alkali Metals – Highly reactive (don’t drop them in water!). They have 1 valence electron they are desperate to give away.
- Group 17: Halogens – Very reactive nonmetals (like Chlorine). They have 7 valence electrons and are desperate to “steal” one more.
- Group 18: Noble Gases – The “introverts” of the table. They have a full outer shell of electrons, making them extremely stable and unlikely to react with anything.
4. Periodic Trends
The “Periodic” in the name comes from the fact that certain properties repeat at regular intervals. This is known as Periodic Law.
- Atomic Radius: Atoms generally get larger as you move down a group and smaller as you move left-to-right across a period.
- Electronegativity: This measures how “greedy” an atom is for electrons. It generally increases as you move toward the top-right of the table (Fluorine is the greediest).
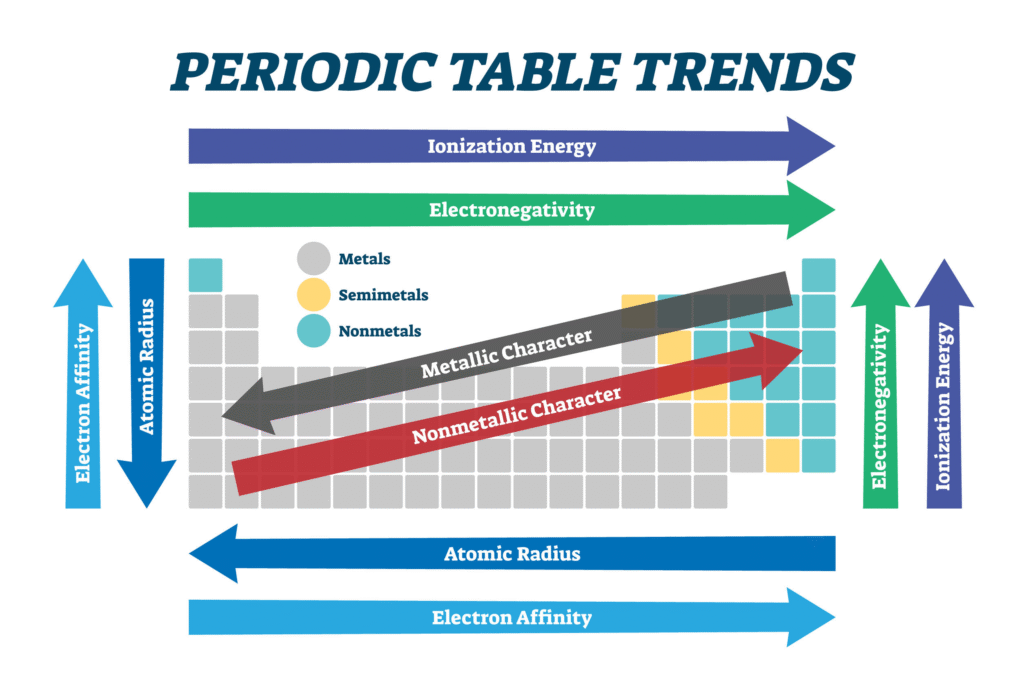
How to Read a Single Tile
Each square on the table provides a “ID card” for that element:
- Atomic Number: Number of protons.
- Symbol: One or two letters (e.g., $Fe$ for Iron).
- Name: The element’s common name.
- Atomic Mass: The weighted average mass of all isotopes.
What is Avagadro’s Constant and how does it lead to the concept of the mole?
Think of Avogadro’s constant as the “baker’s dozen” of the chemistry world—just on a much, much larger scale. It is the bridge between the tiny world of atoms and the human-sized world of the laboratory.
What is Avogadro’s Constant?
Avogadro’s constant (NA) is the number of constituent particles (usually atoms or molecules) found in one mole of a substance.
NA ≈ 6.02214076 * 1023 mol-1
To give you a sense of that scale: if you had a mole of marbles, they would cover the entire surface of the Earth to a depth of about 50 miles. It is a staggering number, but it’s necessary because atoms are incredibly small.
How it Leads to the “Mole”
Before we had advanced technology, scientists knew that different elements reacted in specific ratios by weight, but they couldn’t count individual atoms. They needed a way to talk about “heaps” of atoms that were easy to measure on a scale.
1. The Link to Atomic Mass
The mole was originally defined based on carbon-12. If you took exactly 12 grams of pure carbon-12, the number of atoms inside that pile was designated as “one mole.” Avogadro’s constant is simply the count of those atoms.+1
2. The Great Equalizer
The beauty of this constant is that it allows us to use the Periodic Table effectively.
- An atom of Helium has an atomic mass of approximately 4 u (atomic mass units).
- If you gather one mole (6.022 * 1023 atoms) of Helium, it will weigh exactly 4 grams.
3. Macro vs. Micro
The mole acts as a conversion factor. It allows chemists to transition from the microscopic scale (calculating how many molecules are needed for a reaction) to the macroscopic scale (weighing out grams of powder in a beaker).
| Scale | Unit | Example |
| Microscopic | Atomic Mass Unit (u) | 1 water molecule ≈ 18 u |
| Macroscopic | Grams (g) | 1 mole of water ≈ 18 g |
| The Bridge | Avogadro’s Number | 6.022 * 1023 |
Why does this matter?
Without this concept, chemistry would be guesswork. Because of Avogadro, we know that if we want to react Hydrogen and Oxygen to make water perfectly without any leftovers, we don’t need equal weights of them; we need a specific ratio of moles.
How can the mole concept be used applied in calculations?
Applying the mole concept is all about using conversion factors to move between mass, particle count, and (for gases) volume. Think of the mole as the “central station” in a transit map; to get from Mass to Particles, you must pass through Mole Station first.
The “Mole Map” Logic
To master these calculations, you generally only need two main “tools” from your chemistry toolkit:
- The Periodic Table: To find the Molar Mass (M) of an element or compound.
- Avogadro’s Constant (NA): To convert between moles and the number of particles.
1. Converting Mass to Moles
This is the most common calculation in a lab. If you know how many grams of a substance you have, you can find out how many moles that represents using the formula:
n = m / M
- n = number of moles (mol)
- m = mass of the substance (g)
- M = molar mass (g/mol)
Example: You have 36 grams of water (H2O). The molar mass of water is approximately 18 g/mol (1.0 * 2 + 16).
n = 36 / 18 = 2 moles
2. Converting Moles to Particles
Once you have the number of moles, you can find the exact number of atoms or molecules using Avogadro’s constant.
N = n * NA
- N = total number of particles
- n = number of moles
- NA = 6.022 * 1023
Example: How many molecules are in those 2 moles of water?
N = 2 * (6.022 * 1023) = 1.204 * 1024 molecules
3. Stoichiometry (The “Recipe” Calculation)
The mole concept is most powerful when looking at chemical equations. Equations tell you the mole ratio, not the mass ratio.
Consider the combustion of methane:
CH4 + 2O2 → CO2 + 2H2O
The coefficients tell us that 1 mole of methane requires 2 moles of oxygen. If you have 5 moles of methane, you know instantly you need 10 moles of oxygen. You can then convert those 10 moles back into grams to see exactly how much oxygen gas you need to pump into the reaction.
Summary Table for Quick Reference
| To find… | You need… | Operation |
| Moles | Mass and Molar Mass | Mass / Molar Mass |
| Mass | Moles and Molar Mass | Moles * Molar Mass |
| Number of Particles | Moles | Moles * (6.022 * 1023) |
| Moles | Number of Particles | Particles / (6.022 * 1023) |
