Earth and Atmospheric Sciences
Think of electric forces and electric fields as the “push” and the “map” of the invisible electrical world. They are the fundamental reasons why your hair sticks to a balloon or why your phone charger works.
1. Electric Forces: The “Push and Pull”
An electric force is the actual physical interaction between two charged objects. Much like gravity pulls on mass, electric force pulls or pushes on charges.+1
The behavior of this force is governed by Coulomb’s Law, which tells us two main things:
- Opposites Attract: A positive charge and a negative charge pull toward each other.
- Likes Repel: Two positive charges (or two negative charges) push each other away.
The strength of this force depends on how much charge the objects have and how far apart they are. Mathematically, it looks like this:
F = k |q1 q2| / r2
Where:
- F is the force.
- q1 and q2 are the amounts of charge.
- r is the distance between them.
- k is Coulomb’s constant.
2. Electric Fields: The “Influence Zone”
If the electric force is the action, the electric field is the environment that allows that action to happen.
An electric field is a region of space around a charged object where other charges will feel a force. You can think of it as a “aura” or a “grid” that a charge creates around itself.
How we visualize them:
We use Field Lines to map out this invisible influence:
- Direction: Lines always point away from positive charges and toward negative charges.
- Strength: The closer the lines are to each other, the stronger the field is in that spot.
The Peer-to-Peer Perspective: If you walk into a room and smell cookies, the “smell” is like the field—it’s everywhere in the room whether you eat a cookie or not. The “force” only happens when you actually pick up a cookie and eat it. Similarly, a field exists around a proton even if there isn’t another charge nearby to feel it.
The Key Difference
| Feature | Electric Force (F) | Electric Field (E) |
| Definition | The actual push or pull felt by a charge. | The property of space created by a charge. |
| Requirement | Needs two charges to exist. | Needs only one charge to exist. |
| Analogy | The “weight” you feel holding a rock. | The “gravity” of the Earth itself. |
The relationship between the two is simple: the force (F) felt by a charge (q) in a field (E) is F = qE.
What is the origin of electricity?
To understand the origin of electricity, we have to zoom in past what the eye can see and look at the building blocks of matter: atoms.
Electricity isn’t something humans “invented”; it is a fundamental property of the universe that stems from the subatomic particles that make up everything around us.
1. The Atomic Source
Every atom contains three main particles, but electricity is primarily concerned with two of them:
- Protons: Located in the center (nucleus) with a positive (+) charge.
- Electrons: Orbiting the nucleus with a negative (-) charge.
In a “normal” state, an atom has an equal number of protons and electrons, making it electrically neutral. However, electrons are much lighter than protons and live on the outside of the atom. Because of this, they can be nudged, pulled, or traded between atoms.+2
The “Origin” moment: When an outside force (like friction, magnetism, or a chemical reaction) causes electrons to move from one atom to another, you create an imbalance. This movement and accumulation of charge is the root of all electricity.
2. Static vs. Current: Two Sides of the Same Coin
Depending on how those electrons behave, we categorize electricity into two main types:
Static Electricity (Charges at Rest)
This happens when electrons are “stripped” off one surface and pile up on another.
- Example: When you rub your shoes on a carpet, you are physically scraping electrons off the carpet fibers and onto your body.
- The Spark: You feel a shock when you touch a doorknob because those gathered electrons are jumping all at once to get back to a neutral state.
Current Electricity (Charges in Motion)
This is the “electricity” we use to power our world. It isn’t just a single jump; it’s a steady flow of electrons through a conductor (like a copper wire).
- The Analogy: If static electricity is like a puddle of water, current electricity is like a flowing river.
3. How do we “make” it today?
Since electricity is a natural property, we don’t “create” it from nothing; we convert other forms of energy into electrical energy by forcing electrons to move.
- Magnetism (Generators): Most of our power comes from spinning a coil of wire inside a magnetic field. The magnets “push” the electrons through the wire.+1
- Chemistry (Batteries): Inside a battery, a chemical reaction creates a “surplus” of electrons at one end and a “shortage” at the other. When you plug it in, the electrons flow through your device to reach the other side.
- Light (Solar Panels): Photons from the sun hit a silicon cell and knock electrons loose, creating a flow.
Summary of the “Chain of Origin”
- Matter is made of atoms.
- Atoms contain charged particles (electrons).
- Energy (heat, light, or motion) knocks those electrons loose.
- Electricity is the result of those electrons moving or gathering.
What are charged particles and what are the electric forces that they exert?
To understand electric forces, we have to look at the “social life” of subatomic particles. Charged particles are the source of all electrical phenomena, and the forces they exert are the rules by which they interact.
1. What are Charged Particles?
At the most basic level, a charged particle is a tiny unit of matter that possesses an electric charge. In our everyday world, this almost always refers to the components of an atom:
- Protons: Found in the nucleus; they have a positive (+) charge. They are heavy and generally stay put.
- Electrons: Orbit the nucleus; they have a negative (-) charge. They are light and are the “travelers” that move to create electricity.
- Ions: These are entire atoms that have gained or lost electrons. If an atom loses an electron, it becomes a positive ion; if it gains one, it becomes a negative ion.+1
The Property of “Charge”
Charge is a fundamental property, like mass. Just as mass tells an object how to respond to gravity, charge tells a particle how to respond to electromagnetic fields. We measure this charge in Coulombs (C).
2. The Electric Forces They Exert
When two charged particles get near each other, they exert a force. This isn’t a physical touch; it’s a push or pull that acts through space.+1
The Fundamental Rules of Interaction
The behavior of these forces follows two simple “social” rules:
- Like charges repel: Two protons will push away from each other; two electrons will do the same.
- Opposite charges attract: A proton and an electron will pull toward each other.
What determines the “Strength” of the force?
The force between two particles isn’t constant. According to Coulomb’s Law, two factors change the intensity:
- The Amount of Charge: The more “charged” the particles are, the stronger the push or pull.
- The Distance: This is the most critical factor. The force follows an inverse-square law. If you double the distance between two electrons, the repulsive force between them doesn’t just cut in half—it drops to one-fourth of its original strength.+1
3. Why don’t we feel these forces all the time?
You might wonder: “If my body is made of trillions of protons and electrons, why am I not constantly flying toward a refrigerator or repelling my friends?”
The reason is equilibrium. Most matter is electrically neutral. For every positive proton in your body, there is usually a negative electron canceling it out. The forces are still there at the atomic level, holding your molecules together, but on a large scale, they perfectly balance out to zero.
A Quick Fact: The electric force is incredibly strong compared to gravity. The electric repulsion between two protons is about $10^{36}$ times stronger than the gravitational attraction between them!
What’s the difference between conductors and insulators?
The fundamental difference between a conductor and an insulator comes down to how much “freedom” their electrons have. Think of it like a crowded hallway: in a conductor, the doors are open and everyone is moving freely; in an insulator, everyone is locked in their individual rooms.
1. Conductors: The “Electron Superhighways”
Conductors are materials that allow electricity (electrons) to flow through them easily.
- The Science: In metals, the outer electrons are not tightly bound to their parent atoms. Instead, they form a “sea of electrons” that can drift freely from one atom to another.+1
- Best Examples: Silver, Copper, Gold, Aluminum, and Iron.
- Common Use: This is why your charging cables and household wiring are made of copper—it offers very little “resistance” to the moving charges.
2. Insulators: The “Electric Dead-Ends”
Insulators are materials that prevent or significantly slow down the flow of electricity.
- The Science: In these materials, the electrons are gripped tightly by their atoms. They don’t have enough energy to break free and move, so an electric current can’t easily push through.
- Best Examples: Rubber, Glass, Plastic, Wood, and Air.
- Common Use: This is why those same copper wires are wrapped in plastic or rubber—it keeps the electricity trapped inside the wire and prevents it from shocking you.
3. Comparison at a Glance
| Feature | Conductors | Insulators |
| Electron Mobility | High (free to move) | Low (fixed in place) |
| Resistance | Very Low | Very High |
| Heat Transfer | Usually good heat conductors | Usually good heat insulators |
| Purpose | To carry electrical energy | To block/protect from energy |
4. The “Middle Ground”: Semiconductors
There is actually a third category called semiconductors (like Silicon). These materials are special because they aren’t great conductors or great insulators on their own. However, by adding small impurities or applying a bit of voltage, we can “flip a switch” to make them conduct.
The Peer-to-Peer Perspective: Semiconductors are the “brains” of the modern world. Every computer chip, smartphone, and LED light relies on the ability to precisely control when a material acts like a conductor and when it acts like an insulator.
Is there a relationship between charging by contact and by induction?
Yes, there is a direct relationship: both are methods of transferring or redistributing charge between objects, but they achieve this through different physical mechanisms.
Think of charging by contact as “sharing by touching” and charging by induction as “scaring into moving.”
1. The Core Relationship: Conservation of Charge
Whether you are using contact or induction, both follow the Law of Conservation of Charge. You aren’t “creating” new electricity; you are simply moving electrons from one place to another.
Charging by Contact (Conduction)
This involves physical touch. When a charged object touches a neutral conductor, electrons flow directly from one to the other to balance things out.+1
- The Result: The neutral object gains the same charge as the charging object.
- Permanence: The object remains charged after you pull the source away because the electrons physically jumped across the gap.
2. Charging by Induction
This involves no physical touch. You bring a charged object near a neutral one, which “scares” the electrons in the neutral object to one side.+1
- Polarization: First, the electrons shift. If you bring a negative rod near a sphere, the sphere’s electrons run to the far side.
- Grounding: To make the charge permanent, you must “ground” the object (touch it with a wire or your finger) to let the fleeing electrons escape the object entirely.
- The Result: The neutral object ends up with the opposite charge of the charging object.
3. Comparison of the Two Methods
| Feature | Charging by Contact | Charging by Induction |
| Physical Contact | Required | Not allowed (until grounding) |
| Electron Transfer | Direct jump between objects | Movement within the object (then to ground) |
| Final Charge | Same as the charging source | Opposite of the charging source |
| Material Requirement | Works best with conductors | Requires a conductor and a “ground” |
4. How They Work Together
In many real-world scenarios, these two processes happen in a sequence.
The “Static Shock” Example: > 1. As you walk toward a metal doorknob, your hand (which is charged) induces a charge separation in the metal knob (Induction).
2. Once you get close enough, the air “breaks down” and a spark jumps between your finger and the knob (Contact).
3. You have now charged the knob by contact, and both you and the knob are more neutral than you were a second ago.
The Crucial “Grounding” Connection
The biggest link between the two is the human body or the Earth. In induction, you usually need a “contact” step (grounding) to make the charge permanent. Without that brief moment of contact to let electrons escape or enter, the object would just go back to being neutral the moment you moved the charging rod away.
What is Coulomb’s Law?
Think of Coulomb’s Law as the “Universal Law of Gravitation,” but for electricity. While gravity only pulls things together, Coulomb’s Law describes how charged objects can either pull together or push apart.
Formulated by Charles-Augustin de Coulomb in the 1780s, it provides the mathematical formula to calculate the exact amount of force between two stationary electrical charges.
1. The Core Equation
The law states that the electrical force (F) between two point charges is directly proportional to the product of the charges and inversely proportional to the square of the distance between them.
F = k |q1 q2| / r2
Breaking Down the Variables:
- F (Force): Measured in Newtons (N). This is the “push” or “pull.”
- k (Coulomb’s Constant): A massive number (8.99 * 109, N * m2 / C2). This shows just how much stronger electricity is than gravity.
- q1 and q2 (Charges): Measured in Coulombs (C). The more charge you have, the stronger the force.
- $r$ (Distance): Measured in meters (m). This is the distance between the centers of the two charges.
2. The Two “Golden Rules” of the Law
Coulomb’s Law tells us two very specific things about how the world works:
Rule A: The “Product” Relationship
If you increase the amount of charge on either object, the force increases.
- Double one charge? The force doubles.
- Double both charges? The force becomes 4 times stronger.
Rule B: The “Inverse-Square” Relationship
The distance (r) is squared and sits at the bottom of the fraction. This means the force weakens very quickly as you move charges apart.
- Double the distance (2r)? The force drops to 1/4 (1/22).
- Triple the distance (3r)? The force drops to 1/9 (1/32).
3. Direction Matters: Vector Force
Unlike gravity, which is always attractive, the electric force has a direction based on the signs of the charges:
- Positive (+) Result: If both charges are the same (both + or both -), the force is repulsive. They push away.
- Negative (-) Result: If the charges are opposites (+ and -), the force is attractive. They pull together.
4. Why Does This Matter?
Coulomb’s Law is the reason why atoms stay together. The positive nucleus pulls on the negative electrons with exactly the force calculated by this law. Without this specific mathematical “grip,” matter as we know it would simply fly apart.
Peer Perspective: It’s helpful to remember that k is a constant of the medium. In a vacuum, it’s at its strongest. If you put those same charges in water, the force actually drops significantly because the water molecules get in the way!
What is the electric field?
To understand the electric field, it helps to think of it as an invisible “map of influence.” While the electric force is the actual push or pull you feel, the electric field is the reason that push or pull is possible in the first place.
1. Defining the Field
An electric field (E) is a region of space around a charged object where other charges will experience a force.
If you place a positive proton in an empty room, it “warps” the space around it. Even if there are no other charges nearby to feel it, the field still exists. It’s like the smell of a candle: the scent is in the air (the field) whether or not someone is there to smell it (the force).
2. Visualizing Field Lines
Since we can’t see electric fields, we use field lines to map them out. These lines follow two strict rules:
- Direction: The lines always point away from positive charges and toward negative charges. (This is based on what a tiny “test” positive charge would do if dropped into the field).
- Density: Where the lines are packed tightly together, the field is strong. Where they spread out, the field is weak.
3. The Math: Strength of the Field
The strength of an electric field at any point is defined as the amount of force (F) exerted per unit of charge (q).
E = F / q
Using Coulomb’s Law, we can also calculate the field created by a single “source” charge (Q) at a specific distance (r):
E = k Q / r2
- E: Electric Field strength (measured in Newtons per Coulomb, N/C).
- k: Coulomb’s constant.
- Q: The charge creating the field.
- r: The distance from that charge.
4. Key Properties to Remember
- It’s a Vector: Electric fields have both a magnitude (how strong they are) and a direction (where they point).
- Superposition: If you have multiple charges, their fields don’t “crash” into each other; they simply add up. The total field at any point is the sum of all individual fields present.
- Conductors: Inside a perfect conductor (like a solid metal ball) in equilibrium, the electric field is always zero. All the charges move to the surface to cancel the internal field out.
The Big Picture
The electric field is the “middleman” of physics. Instead of saying “Charge A pulls Charge B,” physicists prefer to say:
- Charge A creates an electric field.
- Charge B interacts with that field and feels a force.
What are electric field lines?
Think of electric field lines as a visual map that helps us “see” the invisible influence surrounding a charged object. Since we can’t see the electric field itself, these lines represent the path a tiny, positive “test charge” would take if it were dropped into the field.
The concept was pioneered by Michael Faraday, who wanted a way to visualize the “lines of force” in space.
1. The Three Golden Rules of Field Lines
To draw or interpret these maps accurately, there are three universal rules you must follow:
- Direction: Lines always point away from positive charges and toward negative charges.
- No Crossing: Field lines never cross each other. If they did, a particle at the intersection wouldn’t know which direction to move!
- Density equals Strength: Where the lines are packed tightly together (close to the charge), the field is strong. As the lines spread out, the field becomes weaker.+1
2. Common Field Patterns
The “shape” of the field depends entirely on the arrangement of the charges.
The Dipole (Opposites)
When a positive and negative charge are near each other, the lines loop directly from the positive to the negative. This creates a strong field in the space between them.
Like Charges (Repulsion)
If you put two positive charges together, the lines push away from each other, leaving a “dead zone” in the exact middle where the electric field is zero.
Parallel Plates (Uniform Field)
When you have two flat metal plates—one positive and one negative—the lines between them are perfectly straight, parallel, and evenly spaced. This is called a uniform electric field, meaning the strength is the same everywhere between the plates.+1
3. Why do we use them?
Beyond just being a handy drawing, field lines tell us two critical things about any point in space:
- The Tangent: The direction of the force on a positive charge at any point is tangent to the field line at that spot.
- The Magnitude: By counting how many lines pass through a specific area (line density), we can calculate the exact intensity of the electric field ($E$).
Peer Perspective: A common mistake is thinking the lines are “real” physical strings in space. They aren’t! They are just a mathematical tool. If you moved a charge slightly, the entire “map” of lines would shift instantly to reflect the new reality.
What is the electric field inside a conductor and what is this called?
The electric field inside a conductor in electrostatic equilibrium is zero. Even if you place a metal object inside a massive external electric field, the field inside the actual material of the conductor remains null.
This phenomenon is known as Electrostatic Shielding (or sometimes the Faraday Cage effect).
1. Why is it Zero?
To understand this, we have to look at the behavior of free electrons within the conductor:
- External Field Applied: Imagine a solid block of copper placed between a positive and a negative plate. An external electric field (Eext) is created between those plates.
- Electron Migration: The free electrons in the copper feel a force and immediately rush toward the positive plate. This leaves the other side of the block with a net positive charge.
- Induced Internal Field: This separation of charges creates its own “internal” electric field (Eint) that points in the opposite direction of the external one.
- The Balance: Electrons will keep moving until the internal field perfectly cancels out the external field. At that point, the net force on the electrons is zero, they stop moving, and the net electric field inside is zero.
2. Key Properties of Shielding
Because the internal field is zero, several interesting things happen:
- Surface Charge: All excess charge sits on the outer surface of the conductor. It never stays in the middle.
- Perpendicular Lines: Electric field lines outside the conductor always hit the surface at a perfect 90° angle.
- Hollow vs. Solid: This works whether the conductor is a solid chunk of metal or a hollow shell (like a birdcage). The space inside the hollow shell is protected from the external field.
3. Real-World Applications: The Faraday Cage
The most famous application of this principle is the Faraday Cage. Because the interior of a conductor has no electric field, anything inside is shielded from outside electrical interference or high-voltage discharges.
- Airplanes: If a plane is struck by lightning, the metal “skin” of the aircraft acts as a conductor. The electricity stays on the outside surface and passes through to the air, leaving the passengers and electronics inside safe.
- Microwave Ovens: The metal mesh on the door of your microwave acts as a shield to keep the electromagnetic fields inside the oven so they don’t cook you while you watch your food.
- Electronic Cables: High-quality audio or data cables are “shielded” with a foil or braided wire wrap to prevent outside electrical noise from distorting the signal.
A Quick Note on “Equilibrium”
It is important to remember this only applies to electrostatic equilibrium (when charges aren’t flowing). If you have a battery hooked up to a wire, you do have an electric field inside the wire—that’s what keeps the current moving!
What is Gauss’ Law?
Think of Gauss’ Law as a more powerful, “big picture” version of Coulomb’s Law. While Coulomb’s Law tells you the force between two specific points, Gauss’ Law relates the electric field on a closed surface to the total charge trapped inside that surface.
It is one of the four Maxwell’s Equations, which form the foundation of all classical electromagnetism.
1. The Concept: Flux and Enclosure
To understand Gauss’ Law, you need to understand Electric Flux ($\Phi_E$). Imagine the electric field lines coming out of a charge are like water spraying from a sprinkler. Flux is a measurement of how much “spray” passes through a given area.
Gauss’ Law states that the total electric flux coming out of any closed surface (an imaginary 3D bubble called a “Gaussian Surface”) is proportional to the net electric charge enclosed inside it.
2. The Equation
In its mathematical form, Gauss’ Law is written as:
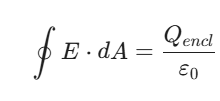
What do these symbols mean?

In plain English: If you add up all the electric field lines “poking through” a bubble, that total is exactly equal to the amount of charge inside divided by a constant.
3. Why is Gauss’ Law so useful?
While Coulomb’s Law is great for simple points, it becomes a nightmare to calculate for complex shapes like long wires or large flat sheets. Gauss’ Law makes these calculations easy if the object has symmetry.
The Three “Symmetries”:
- Spherical Symmetry: Used for point charges or solid metal spheres. You imagine a spherical “bubble” around them.
- Cylindrical Symmetry: Used for long wires or pipes. You imagine a “soda can” bubble around the wire.
- Planar Symmetry: Used for big flat plates (like in a capacitor). You imagine a “pillbox” bubble poking through the plate.
4. Key Takeaways
- Charges Outside Don’t Count: If a charge is outside your imaginary bubble, its field lines go in one side and out the other. The net flux it contributes is zero. Only the charge inside determines the flux.
- Shape Doesn’t Matter: The total flux through the bubble is the same whether the bubble is a sphere, a cube, or a weird potato shape, as long as it encloses the same amount of charge.
- Proof of Zero Field: Gauss’ Law is the mathematical proof for why the electric field inside a conductor is zero (since any internal “bubble” would enclose zero net charge).
How do copiers and computer printers make use of charge?
Modern copiers and laser printers are essentially “applied electrostatics” machines. They rely on the principle that opposite charges attract to move plastic powder (toner) onto paper with extreme precision.
The process, called xerography, generally follows these five steps:
1. Charging the Drum
Inside the printer is a rotating drum coated with a photoconductive material (like selenium). A wire called a corona wire (or a charge roller) gives the entire surface of this drum a uniform positive charge.
2. Imaging (The “Invisible” Map)
The printer uses a laser (or a copier uses a bright light) to “write” the image onto the drum.
- Because the drum is a photoconductor, the areas hit by light become conductive and lose their charge, leaking it away to the ground.
- The dark areas of your image remain positively charged.
- This creates an invisible “electrostatic image” on the drum.
3. Developing (Applying the Toner)
The printer then coats the drum with toner—a fine, black plastic powder.
- The toner is given a negative charge.
- Because opposites attract, the negative toner particles cling only to the positively charged parts of the drum (the parts the laser didn’t hit).
- The toner “ignores” the discharged areas where the laser did hit.
4. Transferring to Paper
Now the printer needs to get that toner off the drum and onto the paper.
- A sheet of paper is passed under the drum and given a stronger positive charge than the drum itself.
- As the paper passes, it “yanks” the negatively charged toner off the drum and onto the page.
5. Fusing (Making it Permanent)
At this stage, the toner is just sitting on the paper like dust. If you blew on it, the image would disappear.
- The paper passes through the fuser—two hot rollers that apply heat and pressure.
- This melts the plastic toner and presses it into the fibers of the paper. This is why a page feels warm when it first comes out of a laser printer.
Summary of Charges
| Component | Charge | Role |
| Drum Surface | Positive (+) | Holds the “template” of the image. |
| Laser/Light | Neutralizes | Erases the background charge. |
| Toner Powder | Negative (-) | Sticks to the remaining positive template. |
| Paper | Strong Positive (++) | Pulls the toner away from the drum. |
Fun Fact: This is also why laser printers are prone to paper jams when the humidity is very high. Moisture in the air makes the paper more conductive, which interferes with its ability to hold a strong static charge, preventing the toner from transferring correctly!
What is the relationship between the electrostatic force and Newton’s laws of motion?
The relationship between electrostatic force and Newton’s laws of motion is a perfect marriage of “Power” and “Instructions.” Electrostatic force (Coulomb’s Law) provides the “push,” while Newton’s laws provide the rules for how that push turns into movement.
In physics, we say that electrostatic force is the cause, and Newton’s laws describe the effect.
1. Newton’s Second Law (F = ma)
This is the most direct link. When two charges exert a force on each other, that force (F) causes the particles to accelerate (a).
- The Formula: k |q1 q2| / r2 = ma
- The Result: Because electrons have an incredibly tiny mass ($m$), even a small electrostatic force causes them to accelerate at mind-blowing speeds. This is why electronics can react in nanoseconds.
2. Newton’s Third Law (Action-Reaction)
Newton’s Third Law states that for every action, there is an equal and opposite reaction. This applies perfectly to electrostatic forces.
- The Interaction: If a massive glass rod with a positive charge pulls on a tiny electron, the electron pulls back on the glass rod with exactly the same amount of force.
- The Difference: You see the electron fly toward the rod, but you don’t see the rod move toward the electron. This isn’t because the forces are different; it’s because the rod’s mass is so much larger that its acceleration is invisible to the human eye.
3. Newton’s First Law (Inertia)
Newton’s First Law tells us that a charge will stay at rest or keep moving in a straight line unless an external force acts on it.
- In a Vacuum: In an old CRT television, an “electron gun” fires electrons in a straight line. They would travel forever in that line (First Law) if it weren’t for the Electric Fields (Force) used to steer them toward the screen to create an image.
4. Work and Energy
Newtonian mechanics defines Work as a force applied over a distance (W = Fd).
When an electric field moves a charge, it is doing “Work” on that particle. This work converts Electrical Potential Energy into Kinetic Energy. This is exactly how a battery works: it uses stored energy to do the “work” of pushing electrons through a circuit.
Comparison Summary
| Concept | The Electrostatic Part | The Newtonian Part |
| Origin | Charges (q1, q2) create the force. | The force is treated as a vector (F). |
| Motion | The Electric Field (E) sets the stage. | The mass (m) determines how fast it moves. |
| Collision | Repulsion keeps atoms from overlapping. | This provides the “Normal Force” that stops you from falling through the floor. |
The Peer-to-Peer Perspective: If you want a real-world example of this relationship, look at a defibrillator. It stores a massive amount of electrostatic potential energy. When the pads touch a patient, that energy creates a massive force that “pushes” electrons through the heart (Newton’s 2nd Law), physically forcing the heart muscle to contract.
