Meteorology Today Second Canadian Edition
Earth and Atmospheric Sciences
In simple terms, atmospheric energy is the fuel that drives our weather and climate. It represents the total amount of energy stored within the Earth’s atmosphere, primarily derived from the Sun.
Think of the atmosphere as a massive heat engine. It takes in solar radiation, converts it into different forms of energy (like heat and motion), and eventually radiates it back out into space.
The Types of Energy in the Atmosphere
To understand how the “engine” works, we look at four main types of energy:
- Sensible Heat (Internal Energy): This is the energy we feel as temperature. When gas molecules move faster, the temperature rises.
- Latent Heat: This is “hidden” energy. It is absorbed or released during phase changes of water—specifically when water evaporates into vapor or condenses into clouds.
- Potential Energy: This depends on gravity and height. Air that is lifted high into the atmosphere has more potential energy than air at the surface.
- Kinetic Energy: This is the energy of motion. In the atmosphere, we see this as wind. While it’s the most visible form of energy, it actually makes up a very small fraction of the total atmospheric energy.
The Energy Cycle: How It Works
The process follows a specific flow that keeps our planet habitable:
- Absorption: The Earth’s surface and atmosphere absorb solar radiation (shortwave).
- Redistribution: Because the equator receives more sun than the poles, the atmosphere moves air around to try and balance the temperature. This creates high and low-pressure systems.
- Conversion: Potential and internal energy are converted into kinetic energy, which gives us everything from a light breeze to a category 5 hurricane.
- Emission: To stay in balance, the Earth must emit the same amount of energy back into space as longwave (infrared) radiation.
Why It Matters
Understanding atmospheric energy is the “holy grail” for meteorologists. If there is an imbalance—such as more energy being trapped by greenhouse gases than is being released—the atmosphere has more “fuel” to work with. This typically results in more intense storms, altered wind patterns, and rising global temperatures.
What is the relationship between energy and heat?

Think of energy as the “total potential to do work” and heat as the “energy in transit.” While we often use the terms interchangeably in casual conversation, in the world of physics, they have a very specific, dynamic relationship.
1. The Core Distinction
The easiest way to separate them is to look at how they exist:
- Energy (Internal Energy): This is a property a system has. It is the sum of all the microscopic motion (kinetic) and the bonds between molecules (potential) inside an object.
- Heat (Q): This is a process. Heat only exists when energy is moving from one place to another due to a temperature difference. You can’t “contain” heat; you can only “transfer” it.
2. The Flow of Energy
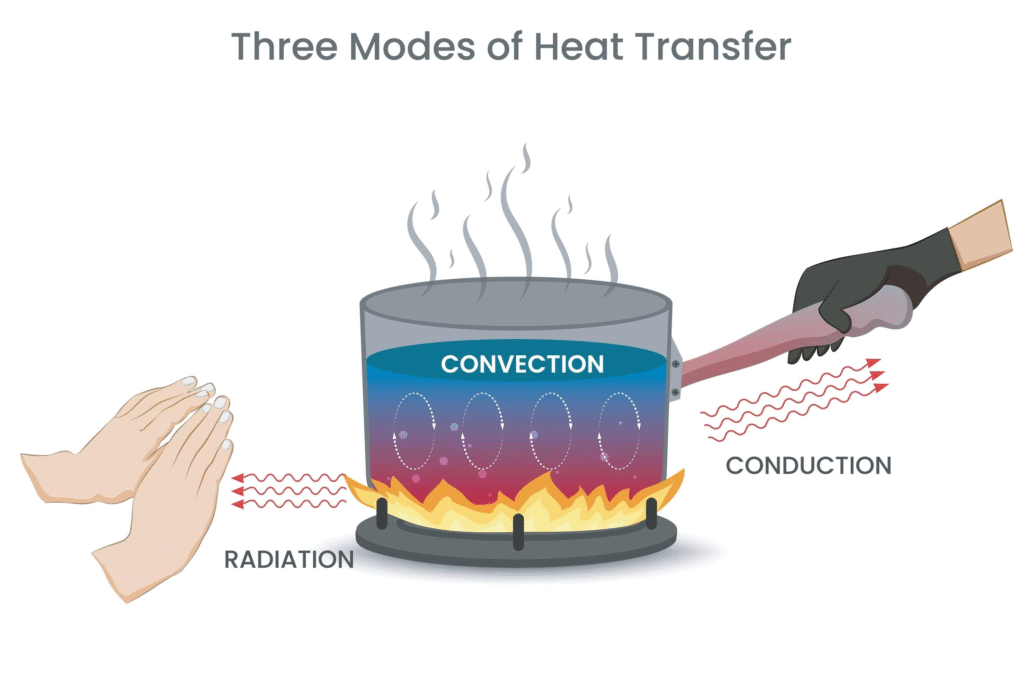
Energy always moves from an area of high concentration (hot) to an area of low concentration (cold). This transfer happens in three ways:
| Method | How it Works | Example |
| Conduction | Direct contact between molecules. | A metal spoon getting hot in a soup bowl. |
| Convection | Movement of fluids (liquids or gases). | Warm air rising above a heater. |
| Radiation | Electromagnetic waves (no medium needed). | Feeling the Sun’s warmth on your skin. |
3. The Formulaic Connection
In thermodynamics, the relationship is often defined by the First Law of Thermodynamics, which states that the change in a system’s internal energy (ΔU) is equal to the heat added to the system (Q) minus the work (W) done by the system:
ΔU = Q – W
This shows that heat is one of the primary ways to change the energy state of an object. If you add heat (Q), the internal energy (ΔU) goes up—unless the system uses that energy to do work (like steam pushing a piston).
4. Heat vs. Temperature
It is a common mistake to think heat and temperature are the same thing.
- Temperature is a measurement of the average kinetic energy of the molecules.
- Heat is the total energy transferred.
The Bathtub Analogy: A cup of boiling water has a much higher temperature than a lukewarm bathtub. However, the bathtub has more internal energy because it has millions more molecules. If you wanted to melt a giant block of ice, the bathtub would be more effective because it can transfer more total heat.
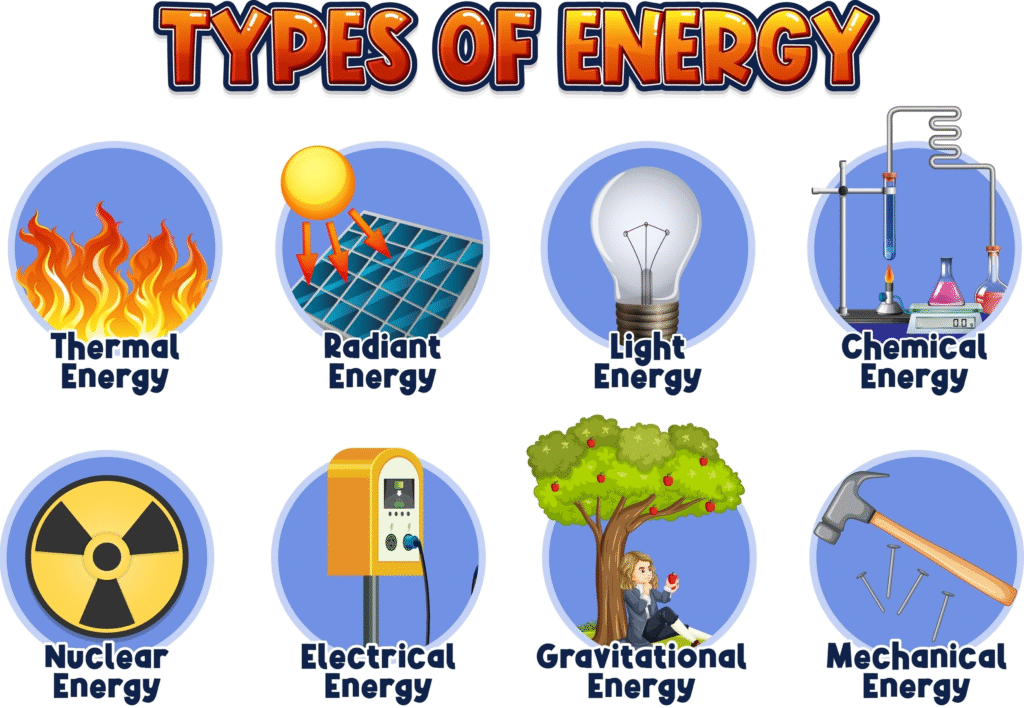
What are the forms of energy?
Energy is never truly created or destroyed; it simply changes from one form to another. Generally, all forms of energy fall into two main categories: Kinetic Energy (the energy of motion) and Potential Energy (stored energy).
Here is a breakdown of the most common forms you’ll encounter:
1. Potential Energy (Stored)
This is energy waiting to be released. It depends on the position or state of an object.
- Chemical Energy: Stored in the bonds of chemical compounds (atoms and molecules). It’s released during a chemical reaction.
- Examples: Food, batteries, gasoline, and wood.
- Mechanical (Elastic) Energy: Stored in objects by the application of a force.
- Examples: A compressed spring or a stretched rubber band.
- Nuclear Energy: Stored in the nucleus of an atom—the energy that holds the nucleus together. It is released when nuclei are combined (fusion) or split (fission).
- Gravitational Energy: Stored in an object’s height. The higher and heavier the object, the more gravitational potential energy it has.
- Example: Water behind a hydropower dam.
2. Kinetic Energy (In Motion)
This is the energy of electromagnetic waves, electrons, atoms, molecules, and substances in motion.
- Radiant (Light) Energy: Electromagnetic energy that travels in transverse waves.
- Examples: Visible light, X-rays, and radio waves.
- Thermal (Heat) Energy: The internal energy in substances—the vibration and movement of atoms and molecules.
- Example: Geothermal energy from the Earth.
- Motion Energy: The movement of objects from one place to another.
- Example: Wind or a moving car.
- Sound Energy: The movement of energy through substances in longitudinal (compression/rarefaction) waves. It is produced when a force causes an object to vibrate.
- Electrical Energy: Delivered by tiny charged particles called electrons, typically moving through a wire.
- Example: Lightning or the electricity in your home.
Energy Transformation
In the real world, energy constantly “shapsifts” between these forms. This is known as energy conversion.
| Device | Energy Start | Energy End |
| Toaster | Electrical | Thermal |
| Flashlight | Chemical (Battery) | Radiant (Light) |
| Car Engine | Chemical (Fuel) | Motion (Kinetic) |
| Solar Panel | Radiant | Electrical |
What are the characteristics of the sun?
The Sun is the powerhouse of our solar system—a nearly perfect sphere of hot plasma that accounts for 99.8% of the total mass in our cosmic neighborhood.
To understand the Sun, it helps to look at its physical properties, its layered structure, and the “behavior” it exhibits through solar activity.
1. Physical Statistics
The Sun is a “Yellow Dwarf” star (G-type main-sequence star). Its scale is difficult to grasp:
- Diameter: About 1.4 million kilometers. You could fit 109 Earths across its face.
- Mass: Roughly 1.989 * 1030 kg, which is about 333,000 times the mass of Earth.
- Composition: Primarily Hydrogen (73%) and Helium (25%), with trace amounts of heavier elements like oxygen, carbon, and iron.
2. Structural Layers
The Sun isn’t solid; it’s organized into distinct layers, each with a different job and temperature.
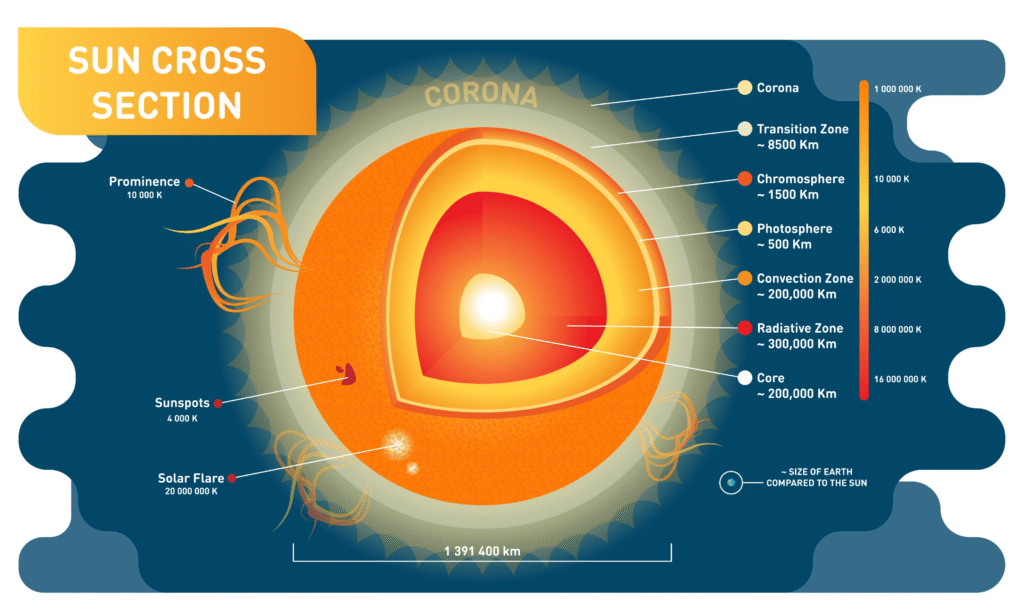
- The Core: The “engine room.” Temperatures reach 15 million°C. This is where nuclear fusion happens, turning hydrogen into helium and releasing massive amounts of energy.
- Radiative Zone: Energy travels outward from the core as radiation. This process is so dense that a single photon can take 170,000 years to bounce its way out to the next layer.
- Convection Zone: Here, hot plasma rises, cools, and sinks in giant loops, much like the movement in a boiling pot of oatmeal.
- Photosphere: The visible “surface” we see from Earth. It’s much cooler, at about 5,500°C.
- Atmosphere: Consists of the Chromosphere and the Corona. The Corona is the outer halo of light visible during an eclipse; strangely, it is much hotter than the surface, reaching millions of degrees.
3. Solar Activity & Magnetism
The Sun is a magnetically active star, which creates several distinct features:
| Feature | What it is |
| Sunspots | Dark, cooler regions on the photosphere caused by intense magnetic activity. |
| Solar Flares | Sudden, massive explosions of energy and radiation. |
| Solar Wind | A constant stream of charged particles flowing outward into the solar system. |
| Prominences | Giant loops of plasma that tether to the surface and extend out into the atmosphere. |
4. The Source of Power: Nuclear Fusion
The most defining characteristic of the Sun is how it generates energy. Unlike a fire that “burns” oxygen, the Sun uses nuclear fusion. Under extreme pressure and heat, hydrogen atoms are crushed together to form helium. This process follows Einstein’s famous equation:
E = mc2
This tells us that a tiny amount of mass is converted into a gargantuan amount of energy, which eventually reaches us as light and heat.
What is sensible heat?
In the simplest terms, sensible heat is heat you can “sense” or feel. It is the energy exchanged between a body and its surroundings that results in a change in temperature without changing the state of the object (like melting or boiling).
When you touch a hot cup of coffee or feel the air get warmer on a summer afternoon, you are experiencing sensible heat.
How It Works
At a molecular level, sensible heat is the kinetic energy of the molecules within a substance.
- As you add sensible heat, the molecules move faster.
- This increase in speed is reflected on a thermometer as a rise in temperature.
Unlike latent heat (which is “hidden” energy used to turn ice into water), sensible heat always shows up as a measurable temperature shift.
The Sensible Heat Formula
In physics and engineering, the amount of sensible heat (Q) required to change the temperature of a substance is calculated using this formula:
Q = mcΔT
Where:
- Q: Sensible heat (Joules)
- m: Mass of the substance (kg)
- c: Specific heat capacity (the “stubbornness” of a material to change temperature)
- ΔT: The change in temperature (Celsius or Kelvin)
Sensible Heat in the Atmosphere
In meteorology, sensible heat is a major player in how energy moves around the planet. It is the process of transferring energy from the Earth’s surface to the atmosphere through conduction and convection.
- Warm Surfaces: The sun heats the ground (sensible heat increases).
- Convection: The air touching the ground warms up, becomes less dense, and rises, carrying that “sensed” heat into the upper atmosphere.
- Thermals: This rising warm air is what glider pilots and birds use to stay aloft.
Sensible vs. Latent Heat: A Quick Comparison
| Feature | Sensible Heat | Latent Heat |
| Effect | Changes Temperature | Changes State (Solid/Liquid/Gas) |
| Measurable? | Yes, with a thermometer | No, temperature stays constant |
| Molecules | Molecules move faster | Molecular bonds are broken/formed |
What is latent heat?
While sensible heat is the energy you feel as a change in temperature, latent heat is “hidden” energy. It is the energy absorbed or released by a substance during a change in its physical state (phase change) that occurs without any change in temperature.
If you put a thermometer in a pot of boiling water, the temperature stays exactly at 100°C even though you are still adding heat from the stove. That “extra” energy is latent heat; it’s being used to break the molecular bonds holding the liquid together rather than making the water hotter.
How Latent Heat Works
Latent heat is all about the “glue” that holds molecules together.
- To go from Solid → Liquid → Gas: You must add energy to break those molecular bonds. This energy is “stored” in the new state.
- To go from Gas → Liquid → Solid: The substance must release that stored energy back into the environment to allow the bonds to reform.
The Two Main Types
There are two primary forms of latent heat that we interact with daily:
- Latent Heat of Fusion: The energy required to change a substance from solid to liquid (melting) or released when it goes from liquid to solid (freezing).
- Latent Heat of Vaporization: The energy required to change a substance from liquid to gas (evaporation/boiling) or released when it goes from gas to liquid (condensation).
Latent Heat in the Atmosphere (The Weather Engine)
This is the most powerful “hidden” force in our weather. Water is the only substance on Earth that exists in all three states (solid, liquid, gas) within normal atmospheric temperatures.
- Evaporation (Storing Energy): As the Sun beats down on the ocean, water evaporates. It “steals” energy from the ocean and stores it in the water vapor.
- Condensation (Releasing Energy): When that vapor rises and cools, it turns back into liquid water to form clouds. At that exact moment, it releases all that stored latent heat into the surrounding air.
Why this matters: This massive release of heat is what fuels thunderstorms and hurricanes. A single hurricane can release energy equivalent to 200 times the entire world’s electrical generating capacity, all driven by the release of latent heat.
Comparison: Heating Curve
If you were to graph the temperature of ice as you heated it until it became steam, it wouldn’t be a straight line. It would look like a set of stairs:
- The Slopes: Sensible heat (Temperature is rising).
- The Flat Plateaus: Latent heat (Temperature is constant while the state changes).
What path does a sunbeam take and what is its fate?
The journey of a sunbeam is a high-speed odyssey that begins in a nuclear furnace and ends as a tiny vibration in an atom on Earth. It is a story of transformation, moving from light to heat and eventually back into the cold void of space.
1. The Birth: The Core to the Surface
A sunbeam starts as a gamma-ray photon created by nuclear fusion in the Sun’s core.
- The Random Walk: It doesn’t just fly out. The Sun is so dense that the photon bounces between atoms for 100,000 to 170,000 years before it finally reaches the surface (the photosphere).
- The Sprint: Once it breaks free, it travels at the speed of light (299,792,458 m/s), reaching Earth in about 8 minutes and 20 seconds.
2. The Atmosphere: The Gauntlet
When the sunbeam hits Earth’s atmosphere, one of three things happens to it:
- Reflection: About 30% of sunbeams hit clouds, ice, or bright sand and are reflected immediately back into space (this is called Albedo).
- Scattering: Some beams hit gas molecules and “shatter,” scattering in different directions. This is why the sky looks blue.
- Transmission: The lucky ones pass straight through to the surface.
3. The Arrival: Absorption and Transformation
Once the sunbeam hits a dark object (like the ocean, a forest, or your asphalt driveway), its journey as “light” ends. It is absorbed.
- Molecular Agitation: The energy from the sunbeam causes the molecules in the object to vibrate faster.
- Conversion to Sensible Heat: The radiant energy becomes thermal energy. The object gets warmer.
4. The Fate: Re-radiation and the Greenhouse Effect
Energy cannot be destroyed, so it must eventually leave. However, it changes its “clothing” before it goes:
- The Transformation: The Earth “breathes” out the energy it absorbed during the day. But it doesn’t send it back as visible light; it sends it back as Longwave Infrared Radiation (heat).
- The Greenhouse Trap: Some of this heat escapes to space, but much of it is absorbed by greenhouse gases (like CO2 and water vapor) and radiated back down. This keeps our planet warm enough for life.
- The Final Exit: Eventually, every bit of energy that arrived as a sunbeam must radiate back out into the vacuum of space to keep the Earth’s energy budget balanced.
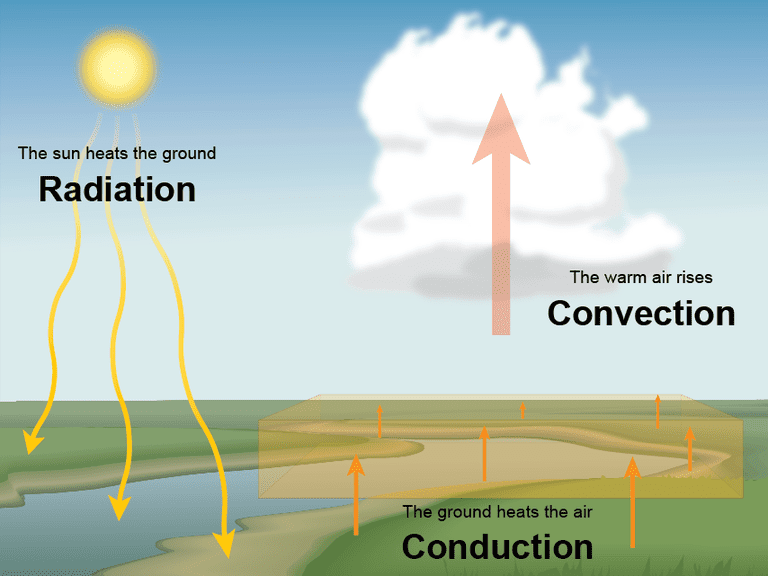
How is heat transferred in the atmosphere?
In the atmosphere, heat is transferred through four primary mechanisms. These processes work together to move energy from the sun-warmed surface into the air and redistribute it from the hot equator toward the cold poles.
1. Radiation (The Initial Source)
Radiation is the transfer of energy via electromagnetic waves. It is the only form of heat transfer that can travel through the vacuum of space.
- Shortwave Radiation: The Sun sends energy to Earth in the form of visible light and UV rays.
- Longwave Radiation: The Earth absorbs this energy and re-emits it as infrared radiation (heat). This is what actually warms the air directly above the ground.
2. Conduction (The Surface Contact)
Conduction is the transfer of heat through direct physical contact.
- Air is a very poor conductor of heat (it’s an insulator).
- Because of this, conduction only happens in a very thin layer of air (just a few centimeters thick) that is touching the Earth’s surface.
- The molecules of the warm ground vibrate rapidly and “bump” into the air molecules, passing on their kinetic energy.
3. Convection (The Vertical Mover)
Convection is the most efficient way heat moves vertically through the atmosphere. It involves the actual movement of a fluid (in this case, air).
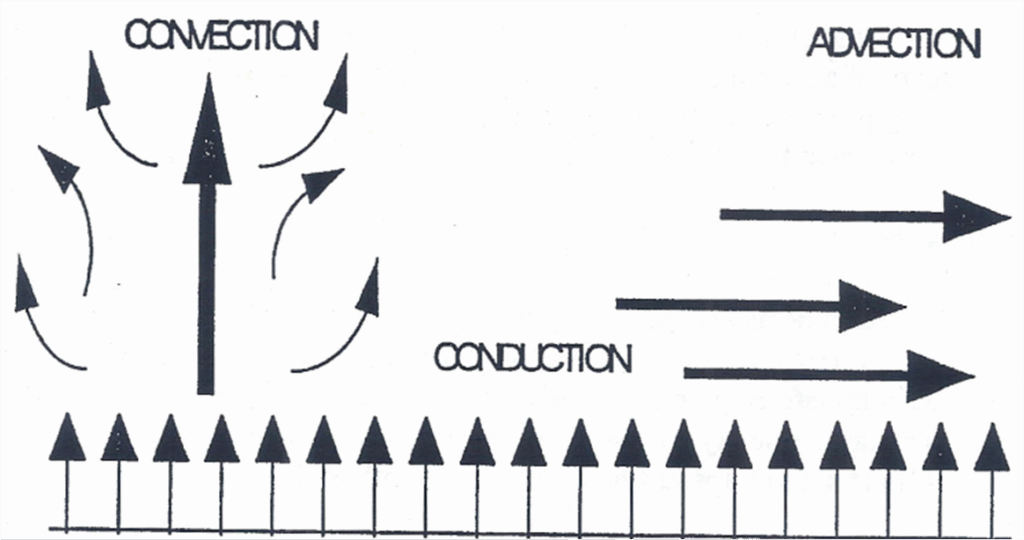
- The Process: As air is warmed by conduction, it becomes less dense and rises (like a hot air balloon). As it rises, it cools, becomes denser, and eventually sinks back down.
- Thermals: These rising loops of warm air are called “convection cells” or thermals. This is how heat from the surface reaches high into the troposphere.
4. Advection (The Horizontal Mover)
While convection moves heat up and down, advection moves heat horizontally across the planet.
- This is primarily driven by wind.
- When a warm air mass moves from the Gulf of Mexico into the northern United States, that is “warm air advection.”
- This is the mechanism responsible for the large-scale redistribution of heat from the tropics to the poles.
Summary Table of Heat Transfer
| Process | Direction | Medium | Real-World Example |
| Radiation | All directions | Waves (No medium) | Feeling the Sun’s warmth on your face. |
| Conduction | Surface to air | Direct contact | Air touching a hot asphalt road. |
| Convection | Vertical (Up) | Moving air | Formation of a thunderstorm cloud. |
| Advection | Horizontal | Wind | A warm breeze blowing from the south. |
What is conduction?
In its simplest form, conduction is the transfer of heat through direct physical contact. It occurs when the microscopic particles (atoms or molecules) of a warmer object collide with the particles of a cooler object, passing along their kinetic energy.
Imagine a “bucket brigade” of energy: the molecules don’t move from one end of the object to the other; they stay in place but vibrate more rapidly, bumping into their neighbors and passing the energy down the line.
How Conduction Works at the Atomic Level
- Agitation: When you heat one end of a solid, the atoms there begin to vibrate more vigorously.
- Collisions: These fast-moving atoms collide with the slower-moving atoms next to them.
- Transfer: During the collision, energy is transferred to the slower atoms, causing them to speed up.
- Chain Reaction: This process continues until the energy is distributed throughout the material or passed to another object touching it.
Conductors vs. Insulators
Not all materials are equally good at conduction. The efficiency depends on how closely packed the atoms are and whether there are “free electrons” to help move the energy.
- Conductors: Materials that transfer heat quickly. Metals (like copper, gold, and aluminum) are excellent conductors because their electrons move freely, acting like “energy couriers.”
- Insulators: Materials that transfer heat very slowly. Wood, plastic, glass, and air are great insulators because their molecules are either far apart or held in rigid structures that prevent easy energy transfer.
Conduction in the Atmosphere
As mentioned earlier, air is a very poor conductor. This is why you can stand near a hot campfire without your skin burning immediately—the air acts as a shield.
However, conduction is vital at the Earth’s surface:
- During the day, the Sun heats the ground (via radiation).
- The ground becomes much hotter than the air.
- A very thin layer of air (only a few centimeters thick) touching the ground warms up through conduction.
- Once that thin layer is warm, convection takes over to lift that heat higher into the sky.
Everyday Examples
- The Spoon in the Soup: A metal spoon sitting in hot liquid will eventually become hot at the handle through conduction.
- Walking on Hot Sand: Your feet feel the heat because of the direct contact and conduction from the sand grains to your skin.
- Ice Melting in Your Hand: Heat moves from your warm skin (high energy) into the cold ice (low energy) until the ice gains enough energy to change state.
What is convection?
Convection is the transfer of heat through the bulk movement of a fluid, such as a liquid or a gas. Unlike conduction (where energy moves through stationary particles), convection involves the warmer, less dense parts of a fluid actually traveling to a new location, carrying their energy with them.
In the atmosphere and the oceans, convection is the primary engine for moving heat vertically.
How Convection Works: The Cycle
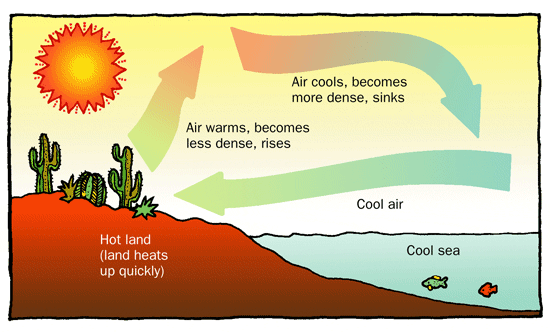
Convection operates in a continuous loop called a convection current. It follows a four-step logic:
- Heating: A fluid (like air or water) is heated from below, causing its molecules to move faster and spread apart.
- Expansion & Buoyancy: Because the molecules are spread out, the fluid becomes less dense (lighter) than the cooler fluid surrounding it.
- Rising: The lighter, warm fluid rises upward.
- Cooling & Sinking: As it moves away from the heat source, it cools, becomes denser (heavier), and sinks back down to be heated again.
Convection in the Atmosphere
This is the process that creates our weather. The Sun doesn’t heat the air directly; it heats the ground, and the ground heats the air touching it.
- Thermals: As the surface warms, bubbles of hot air rise. These are called “thermals.”
- Cloud Formation: As this warm air rises, it cools. If it contains enough moisture, the water vapor condenses to form clouds.
- Thunderstorms: A thunderstorm is essentially a massive, violent convection cell.
Two Types of Convection
In science and engineering, we distinguish between how the fluid starts moving:
| Type | How it Starts | Example |
| Natural (Free) | Driven by buoyancy and temperature differences. | Hot air rising from a toaster. |
| Forced | Driven by an external force like a pump or fan. | A convection oven or a cooling fan in a laptop. |
Real-World Examples
- Boiling Water: You can see the “rolling” motion of the water as the hot liquid at the bottom rises and the cooler liquid at the top sinks.
- Sea Breezes: During the day, the land heats up faster than the ocean. The warm air over the land rises, and the cooler air from the sea rushes in to fill the gap.
- The Earth’s Mantle: Deep beneath our feet, solid rock (acting like a very slow fluid) undergoes convection, which is the force that moves tectonic plates.
What is radiation?
In the context of heat transfer, radiation is the movement of energy through electromagnetic waves.
It is unique among the three types of heat transfer (conduction, convection, and radiation) because it does not require a medium. While conduction needs a solid and convection needs a fluid, radiation can travel through the absolute vacuum of space. Without radiation, life on Earth wouldn’t exist, as it is the only way solar energy can reach us.
1. How Radiation Works
Everything in the universe that has a temperature above “Absolute Zero” (-273.15°C) emits radiation.
- Molecular Vibration: At the atomic level, charged particles (electrons and protons) are constantly moving. This motion creates oscillating magnetic and electric fields that travel outward as waves.
- The Speed: Radiation travels at the speed of light (299,792,458 meters per second).
- Absorption: When these waves hit an object, they can be reflected, transmitted (pass through), or absorbed. When they are absorbed, the energy is converted back into thermal energy, making the object warmer.
2. The Atmosphere’s Radiation Balance
In the atmosphere, we deal with two main types of radiation, defined by their wavelengths:
- Shortwave Radiation (Solar): The Sun is extremely hot, so it emits high-energy, shortwave radiation (mostly visible light and ultraviolet). This passes through the atmosphere relatively easily.
- Longwave Radiation (Terrestrial): The Earth is much cooler than the Sun. It absorbs the shortwave solar energy and re-emits it as lower-energy, longwave infrared radiation.
3. Key Principles of Radiation
Scientists use a few “rules” to predict how radiation behaves:
- The Color Factor (Albedo): Dark-colored objects (like asphalt) absorb most of the radiation that hits them. Light-colored objects (like snow or clouds) reflect most of it.
- The Temperature Factor: The hotter an object is, the more radiation it emits and the shorter its wavelength will be. This is why a “white-hot” piece of metal is hotter than a “red-hot” one.
- The Greenhouse Effect: Greenhouse gases like CO2 and water vapor are “selective absorbers.” They let shortwave solar radiation in but absorb the longwave infrared radiation trying to leave, trapping heat in our atmosphere.
4. Everyday Examples
- A Campfire: Even if the air is cold and you aren’t touching the flames, you feel the heat on your face. That is infrared radiation.
- The Sun: Feeling the warmth of the sun on a cold winter day. The air is cold (poor conduction), but the radiation warms your skin directly.
- A Microwave Oven: It uses specific radio-frequency radiation to flip water molecules back and forth, creating friction and heat inside your food.
What is the relationship between radiation and temperature?
The relationship between radiation and temperature is one of the most fundamental principles in physics. Put simply: the hotter an object is, the more energy it radiates and the shorter its wavelengths become.
This relationship is governed by two primary laws of physics that explain why the Sun looks yellow while a human body “glows” only in invisible infrared.
1. The Stefan-Boltzmann Law: Total Energy
This law describes quantity. It states that the total energy radiated by an object increases very rapidly as its temperature rises. Specifically, the energy (E) is proportional to the fourth power of its absolute temperature (T):
E = σT4
What this means in plain English: If you double the temperature of an object, it doesn’t just radiate twice as much energy—it radiates 16 times as much energy (24 = 16). Small increases in temperature lead to massive increases in the heat energy emitted.
2. Wien’s Displacement Law: Wavelength and Color
This law describes quality (or “color”). It explains that as an object gets hotter, the peak of its radiation shifts toward shorter wavelengths.
- Cooler objects (like a person or a room heater) emit longwave radiation, which we call Infrared.
- Hotter objects (like a stovetop element) begin to emit shorter waves that we can actually see as red light.
- Extremely hot objects (like the Sun) peak in the even shorter visible light spectrum (yellow/white).
3. Real-World Applications
| Object | Temperature | Primary Radiation Type |
| Deep Space | -270°C | Microwaves (very long) |
| Human Body | 37°C | Thermal Infrared (invisible) |
| Incandescent Bulb | 2,200°C | Near-Infrared and Red light |
| The Sun | 5,500°C | Visible Light (short) |
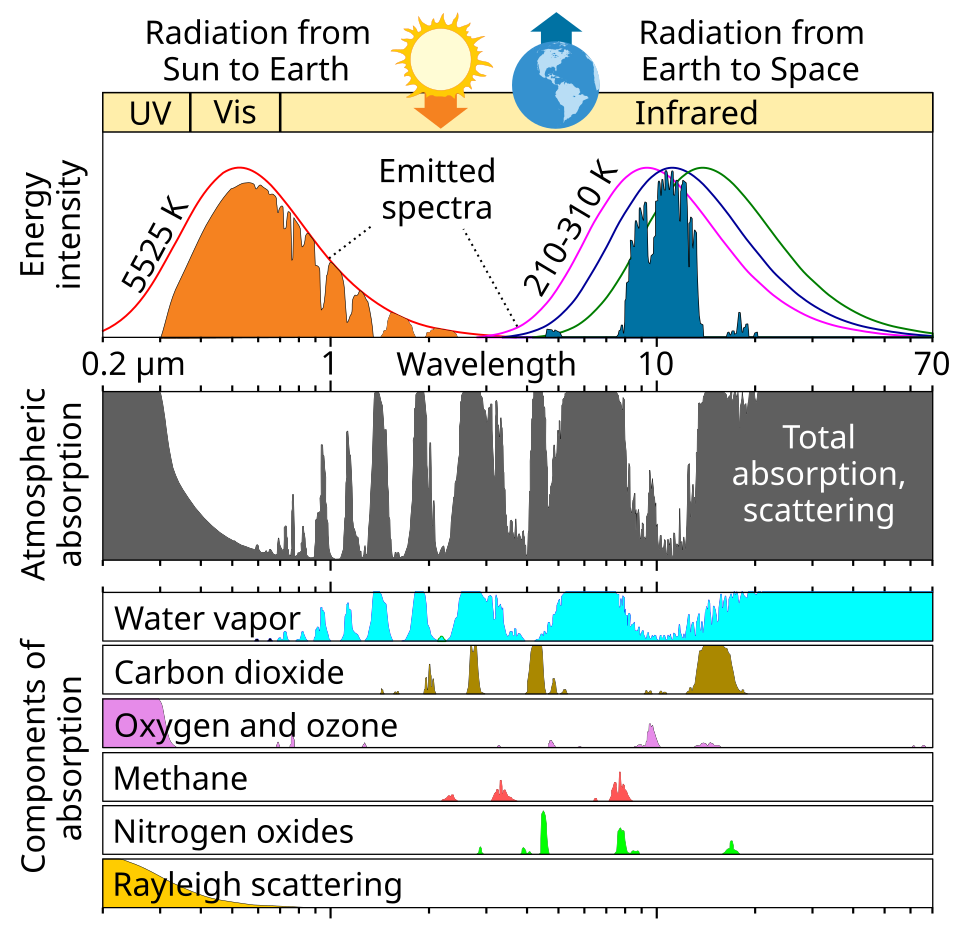
4. The “Atmospheric Window”
This relationship is the reason for the Greenhouse Effect.
- The Sun, being incredibly hot, sends shortwave radiation to Earth. Most of this passes right through the atmosphere like a ghost through a wall.
- The Earth absorbs this energy and warms up, but it only reaches about 15°C on average.
- Because Earth is much cooler than the Sun, it radiates longwave (infrared) energy.
- Greenhouse gases are “calibrated” to block those specific long wavelengths, trapping the heat and keeping the planet warm.
What is the relationship between radiation of the sun and that of the earth?
The relationship between the Sun’s radiation and the Earth’s radiation is a delicate balancing act known as the Earth’s Energy Budget. While both bodies emit radiation, they do so in very different “languages” because of their temperature difference.
The most important takeaway is this: The Sun warms the Earth with shortwave radiation, and the Earth cools itself by emitting longwave radiation.
1. Wavelength: Shortwave vs. Longwave
As we discussed with Wien’s Law, the wavelength of radiation is determined by the temperature of the object.
- The Sun (Shortwave): Because the Sun is extremely hot (roughly 5,500°C), it emits high-energy, shortwave radiation. This includes ultraviolet, visible light, and near-infrared.
- The Earth (Longwave): The Earth is much cooler (averaging 15°C). Therefore, it emits lower-energy, longwave radiation, which falls entirely in the thermal infrared part of the spectrum.
2. The Atmospheric Filter
The atmosphere treats these two types of radiation very differently. This “selective absorption” is what makes life on Earth possible:
- Transparency to Solar: The atmosphere is mostly transparent to incoming shortwave radiation. It allows visible light to pass through and hit the surface (like light through a glass window).
- Opacity to Terrestrial: The atmosphere is “opaque” (blocks) to much of the Earth’s outgoing longwave radiation. Greenhouse gases (CO2, methane, water vapor) absorb this heat and radiate it back down.
3. The Energy Balance
To maintain a stable temperature, the Earth must reach an equilibrium. The amount of energy coming in from the Sun must eventually be equaled by the amount of energy leaving the Earth.
| Direction | Source | Type | Primary Wavelength |
| Incoming | Sun | Solar / Shortwave | Visible Light |
| Outgoing | Earth | Terrestrial / Longwave | Thermal Infrared |
If the Earth emitted less energy than it received, the planet would heat up indefinitely. If it emitted more, it would freeze.
4. Summary of the Relationship
You can think of the relationship as a transformation process:
- Input: The Earth receives high-quality, high-frequency “packets” of light from the Sun.
- Absorption: The Earth’s surface absorbs that light, and the atoms start vibrating (turning light into heat).
- Output: The Earth “exhausts” that heat as low-quality, low-frequency infrared waves.
Crucial Concept: The Earth does not “reflect” the Sun’s heat to stay cool; it converts it. Reflection happens instantly (like light off a mirror), but the radiation of the Earth is a separate process of the planet actually “glowing” in the infrared spectrum.
What is wave energy and what is the relationship between sunburns and ultraviolet rays?
To wrap up our look at atmospheric energy, we need to shift from the large-scale movement of heat to the specific behavior of waves—both in the ocean and in the invisible light spectrum.
1. What is Wave Energy?
Wave energy is a form of kinetic energy caused by the movement of waves across the surface of a liquid (usually the ocean).
- The Source: Most wave energy is actually redirected wind energy. As wind blows across the surface of the water, friction transfers energy from the air to the water, creating ripples that grow into swells.
- The Movement: Interestingly, the water molecules themselves don’t travel very far. They mostly move in a circular motion. It is the energy that moves forward through the water.
- Renewable Power: Engineers use “Wave Energy Converters” to capture this motion and turn it into electricity. Because water is much denser than air, waves carry a massive amount of energy in a small space.
2. The Relationship Between Sunburns and UV Rays
A sunburn isn’t actually a “heat burn” like you’d get from a stove; it is a radiation burn caused by Ultraviolet (UV) rays.
The relationship is all about wavelength and energy levels:
- The UV Spectrum: Ultraviolet radiation has shorter wavelengths and higher frequencies than visible light. Because the wavelengths are so short, they have enough energy to penetrate the outer layer of your skin.
- DNA Damage: When UV rays (specifically UVB) hit your skin cells, the energy is high enough to physically break the bonds of your DNA.
- The Inflammatory Response: A sunburn is your body’s “emergency response” to this DNA damage. Your blood vessels dilate to bring immune cells to the area to clear away the damaged cells, which is why your skin turns red and feels hot to the touch.
- The Tan: Melanin (the pigment in your skin) is your body’s natural “shield.” It absorbs UV rays and turns them into harmless heat before they can hit your DNA. A tan is your body’s way of frantically building more shields after it has already detected damage.
Summary of Solar Radiation Types
To see how these fit together with what we’ve discussed:
| Radiation Type | Effect on You | Role in Atmosphere |
| Infrared | You feel it as Heat. | Warms the air and ground. |
| Visible Light | You use it to See. | Drives photosynthesis and Albedo. |
| Ultraviolet | It causes Sunburns. | Mostly absorbed by the Ozone layer. |
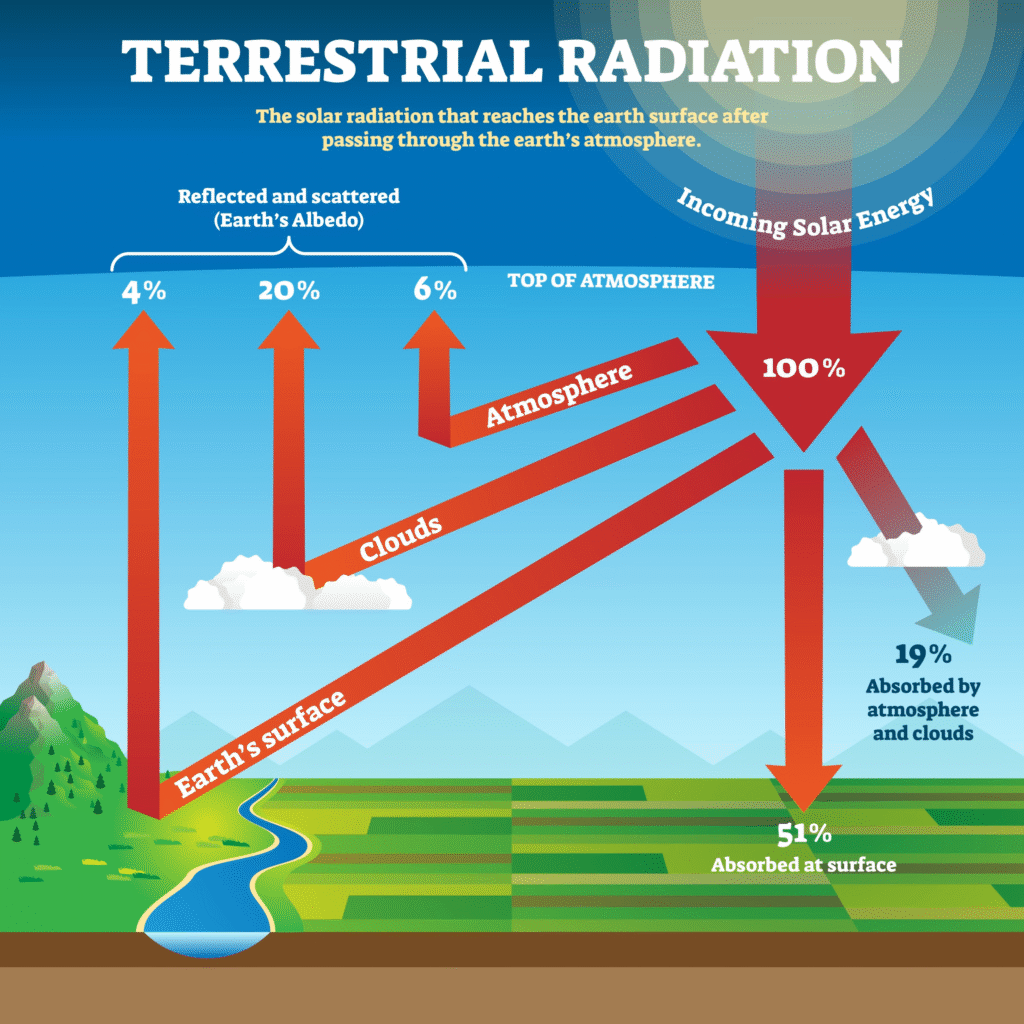
What is incoming solar energy?
Incoming solar energy, often called Insolation (short for Incoming Solar Radiation), is the total amount of solar radiation that reaches a specific area of the Earth’s atmosphere or surface.
It is the primary engine for almost every process on our planet, from the wind and ocean currents to the growth of the food you eat.
1. The Composition of the “Sunbeam”
As we’ve explored, the energy arriving from the Sun isn’t just one thing. It is a “cocktail” of different electromagnetic wavelengths:
- Visible Light (45%): The colors we see.
- Infrared Radiation (45%): The heat we feel.
- Ultraviolet (UV) Radiation (10%): High-energy waves that cause sunburns.
2. The Solar Constant
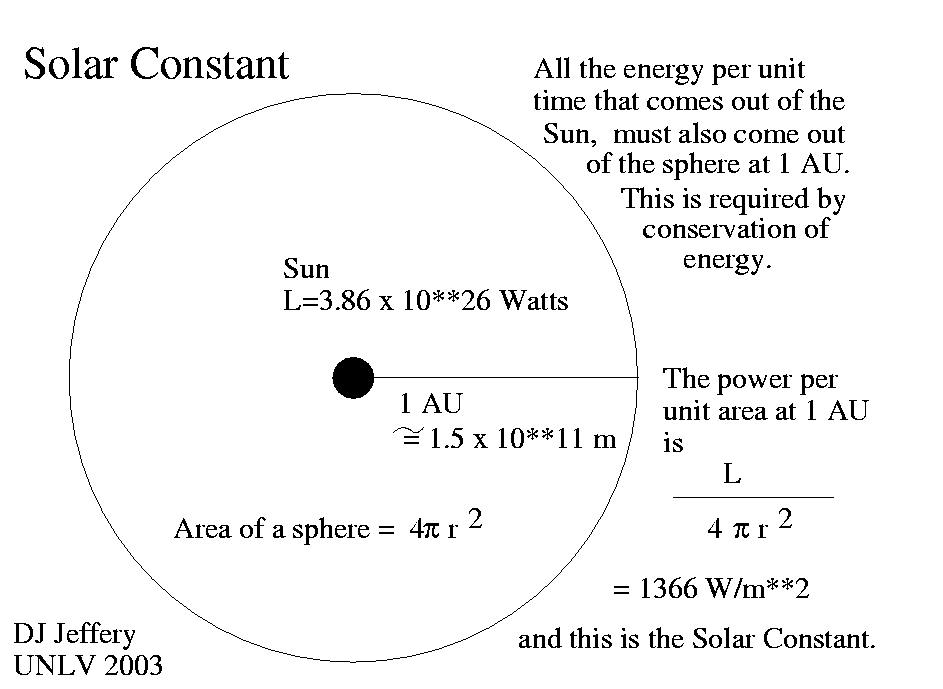
Before the Sun’s energy hits our atmosphere, it is incredibly consistent. Scientists measure this as the Solar Constant.
At the very top of our atmosphere, the Sun delivers approximately 1,361 Watts per square meter (W/m2). If you could put a one-meter by one-meter solar panel in space, it would receive enough power to run about 13 bright old-fashioned lightbulbs continuously.
3. Factors That Influence Incoming Energy
The amount of energy that actually reaches the ground varies wildly based on three main factors:
- The Angle of Incidence: Near the equator, the sun hits the Earth directly (90° angle), concentrating energy in a small area. Near the poles, the same amount of sunlight is spread out over a much larger area because of the Earth’s curve.
- Atmospheric Path Length: Sunlight hitting the poles has to travel through much more “thick” atmosphere than sunlight hitting the equator, meaning more energy is lost or scattered along the way.
- Cloud Cover and Albedo: Clouds act like giant mirrors. On a cloudy day, a huge portion of incoming solar energy is reflected back into space before it ever reaches the surface.
4. The Fate of the Energy
Once the energy arrives, it follows a strict “budget”:
- 30% is Reflected: Immediately bounced back to space by clouds, ice, and the atmosphere.
- 23% is Absorbed by the Atmosphere: Dust, water vapor, and ozone soak up some of the energy.
- 47% is Absorbed by the Surface: This is what warms the oceans and the land.
Why “Insolation” Matters
Understanding incoming solar energy is the key to understanding Climate Zones. Because the tropics receive more consistent, direct incoming energy than the poles, we have a temperature imbalance. The atmosphere’s entire “job” is to try and move that excess energy from the equator to the poles, which is exactly what creates our wind and weather.
What is the difference and the connection between scattering and reflection?
While both reflection and scattering involve light “bouncing” off an object, the difference lies in the direction and surface involved. In the atmosphere, reflection is like a mirror, while scattering is like a disco ball.
1. The Key Differences
The primary distinction is how organized the light remains after it hits a surface.
| Feature | Reflection | Scattering |
| Surface | Occurs on large, smooth, or flat surfaces (clouds, ice, mirrors). | Occurs on tiny particles (gas molecules, dust, water droplets). |
| Direction | Light bounces back at a predictable angle (Angle of Incidence = Angle of Reflection). | Light is sent in all directions (diffuse). |
| Integrity | The light beam stays mostly “whole” and moves in one direction. | The light beam is “shattered” or redirected randomly. |
2. The Connection: They Are Part of “Albedo”
Both reflection and scattering fall under the umbrella of Albedo. Albedo is the measure of how much incoming solar radiation is sent back into space without being absorbed.
- The Shared Goal: Both processes prevent the Earth from getting too hot. Because the energy is redirected away before it can be absorbed, it never turns into sensible heat.
- The Sequence: Often, a sunbeam is scattered by gas molecules in the upper atmosphere, and then the scattered light hits a cloud and is reflected back out into space.
3. Scattering in the Atmosphere
Scattering is why our sky isn’t black. There are two main types you see every day:
- Rayleigh Scattering: This happens when light hits tiny gas molecules (N2 and O2). It favors shorter wavelengths (blue and violet). This is why the sky is blue. At sunset, the light travels through more atmosphere, scattering away the blue and leaving only the long-wave reds and oranges.
- Mie Scattering: This happens when light hits larger particles like water droplets in clouds or smoke. It scatters all wavelengths equally, which is why clouds look white.
4. Reflection in the Atmosphere
Reflection is most common with clouds and ice.
- Cloud Albedo: Thick clouds can reflect up to 90% of the sunlight hitting them, keeping the ground underneath much cooler than it would be on a clear day.
- The Polar Mirror: Snow and ice are highly reflective. This is a critical part of the Earth’s climate; the more ice we have, the more solar energy we reflect back into space.
Summary
If a sunbeam hits a “wall” (like a cloud), it reflects. If it hits a “crowd” of tiny molecules, it scatters. In both cases, the energy is sent away, and the Earth stays a little bit cooler.
Why is the sky blue, sunsets red, and clouds white?
The reason the sky changes color is all due to Scattering, specifically how sunlight interacts with the different sized particles in our atmosphere.
1. Why is the sky blue? (Rayleigh Scattering)
As sunlight enters the atmosphere, it collides with tiny gas molecules (mostly nitrogen and oxygen). These molecules are much smaller than the wavelength of visible light.
- The Selective Bounce: Small molecules are very effective at scattering shorter wavelengths (blue and violet) while letting longer wavelengths (red and yellow) pass through undisturbed.
- The Result: Because blue light is scattered in every direction across the sky, your eyes see blue coming from every part of the atmosphere above you.
- Wait, why not violet? Sunlight contains more blue than violet, and human eyes are much more sensitive to blue.
2. Why are sunsets red? (The Long Path)
At sunrise or sunset, the Sun is low on the horizon. The sunlight has to travel through a much thicker slice of the atmosphere to reach your eyes compared to when it is directly overhead.
- The Filtering Effect: By the time the light reaches you, the atmosphere has scattered away almost all of the blue and violet light.
- What’s Left: Only the longest wavelengths—the reds, oranges, and yellows—are “tough” enough to make it through that long journey without being scattered away.
3. Why are clouds white? (Mie Scattering)
Clouds are made of water droplets and ice crystals, which are much larger than gas molecules.
- The “Equal Opportunity” Scatterer: Because these droplets are large, they don’t care about wavelength. They scatter all colors of the visible spectrum equally in all directions.
- The Mix: When you blend all the colors of light together (red, orange, yellow, green, blue, indigo, violet), you get pure white light.
- Why are some clouds gray? If a cloud is very thick or dense, the light gets scattered so many times that much of it is absorbed or reflected back upward before it can reach the bottom. This leaves the underside of the cloud in “shadow.”
What is radiation absorption, emission, and equilibrium?
To understand how the Earth (or any object) manages its temperature, we look at the three-way relationship between absorption, emission, and equilibrium. This is the fundamental “accounting system” for energy in the universe.
1. Radiation Absorption (Taking Energy In)
Absorption occurs when electromagnetic waves hit an object and the energy is transferred to its molecules.
- What happens: When an atom absorbs a photon (a particle of light), that energy is converted into internal energy. This usually makes the molecules move or vibrate faster, which we measure as an increase in sensible heat (temperature).
- Selectivity: Materials are “picky.” For example, the Earth’s surface is a great absorber of visible light, while greenhouse gases in the atmosphere are poor absorbers of visible light but excellent absorbers of infrared (heat) radiation.
- Dark vs. Light: Darker surfaces have high absorptivity, meaning they soak up more solar energy, while light surfaces reflect it.
2. Radiation Emission (Giving Energy Back)
Emission is the process by which an object sends energy back out into its surroundings.
- The Golden Rule: Every object with a temperature above absolute zero emits radiation.
- The Temperature Link: As we discussed with the Stefan-Boltzmann Law, the hotter an object gets, the more energy it emits.
- The Mechanism: Emission is the Earth’s way of “cooling off.” Without emission, the Earth would simply store every sunbeam it ever received and eventually melt.
3. Radiative Equilibrium (The Balance)
Radiative equilibrium is the state where the rate of absorption equals the rate of emission.
- Steady Temperature: When an object absorbs exactly as much energy as it emits, its temperature remains constant.
- The Earth’s Goal: The Earth is constantly striving for this balance. If the Sun’s intensity increases, the Earth will warm up. As it warms up, it begins to emit more radiation (because hotter things emit more). Eventually, it gets hot enough that its emission matches the new higher absorption level, and a new equilibrium is reached.
The Greenhouse Effect: Shifting the Equilibrium
The current conversation about climate change is essentially a conversation about equilibrium.
- The Trap: Greenhouse gases act like a “blanket,” making it harder for the Earth to emit radiation to space.
- The Imbalance: For a period of time, Absorption > Emission.
- The Adjustment: To get back into equilibrium, the Earth must warm up. A warmer Earth emits more energy. It will continue to get hotter until it is finally “bright” enough in the infrared spectrum to force enough energy through the greenhouse gases to match the incoming solar energy again.
Summary:
- Absorption = Adding to the bank account.
- Emission = Spending from the bank account.
- Equilibrium = A balanced budget where your net worth (temperature) stays the same.
What are selective absorbers and what causes the greenhouse effect?
What are energy balances?
An energy balance is a scientific “accounting” method used to track all the energy entering and leaving a system. According to the Law of Conservation of Energy, energy cannot be created or destroyed, only transformed. Therefore, in any system, the energy that goes in must either be stored, used to do work, or sent back out.
When scientists talk about energy balances, they are usually looking at the Net Energy, which determines whether the system is heating up, cooling down, or staying stable.
1. The Earth-Atmosphere Energy Balance
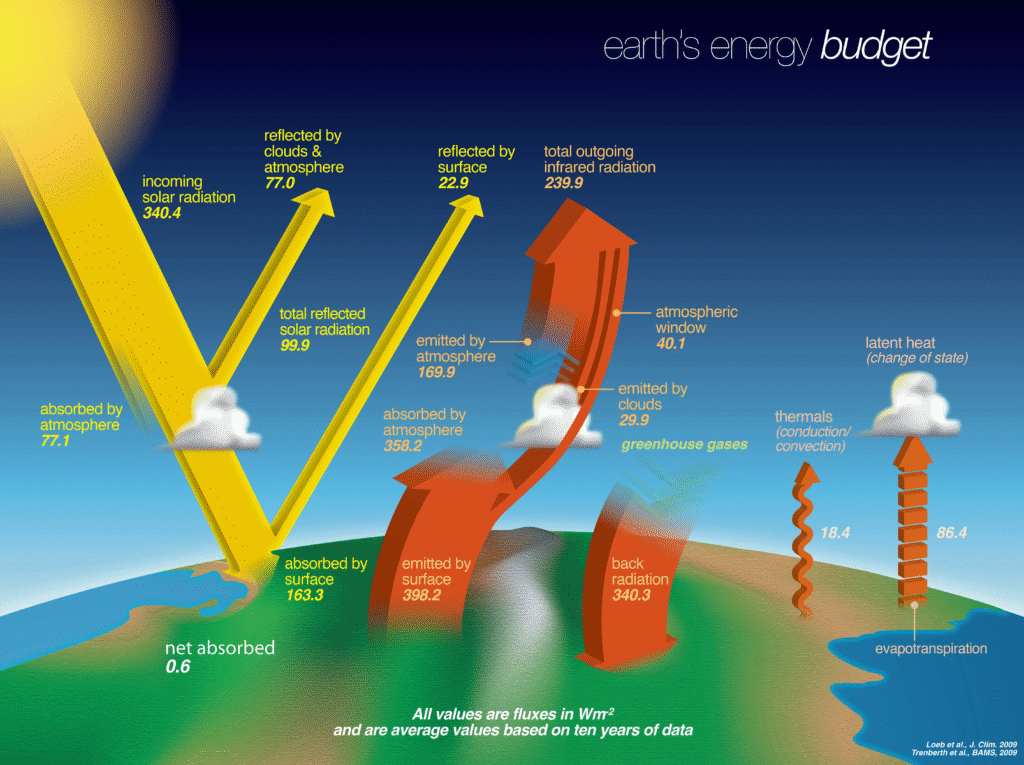
The most famous example is the Global Energy Budget. It tracks the “income” from the Sun and the “expenses” emitted by the Earth back into space.
- Income (Shortwave Radiation): Direct solar energy hitting the top of the atmosphere.
- Expenses (Longwave Radiation & Reflection): * Albedo: About 30% is reflected immediately (by clouds, ice, and dust).
- Outgoing Longwave Radiation (OLR): Heat emitted by the Earth’s surface and atmosphere.
- The Goal (Equilibrium): If Incoming = Outgoing, the Earth’s average temperature remains steady.
2. The Components of a Local Energy Balance
On a smaller scale (like a single forest, a city, or even a leaf), the energy balance is often expressed by the formula:
Q* = H + LE + G
Where:
- Q* (Net Radiation): The total energy available at the surface (Incoming minus Outgoing).
- H (Sensible Heat Flux): Energy used to warm the air.
- LE (Latent Heat Flux): Energy used for evaporation (like sweating or plant transpiration).
- G (Ground Heat Flux): Energy conducted into the soil or pavement.
3. Real-World Applications
Energy balances explain why different environments behave differently:
| Environment | Energy Balance Behavior | Result |
| Deserts | Very low LE (no water), so almost all Q* becomes H. | Temperatures soar during the day. |
| Tropical Rainforests | High LE (lots of plants and moisture). | Much of the energy is used for evaporation, keeping temperatures more stable. |
| Cities | High G (concrete/asphalt soak up heat) and low LE. | This creates the Urban Heat Island effect. |
4. What Happens During an Imbalance?
- Positive Balance (In > Out): The system stores energy. In the case of the planet, this leads to Global Warming. The Earth must eventually heat up until its emission (Out) increases enough to match the In.
- Negative Balance (Out > In): The system loses energy. This happens every night when the Sun goes down; the Earth continues to emit heat but receives no solar income, causing the temperature to drop.
Why does the air warm from below?
It seems counterintuitive—if the Sun is the source of our heat, you would think the air closest to the Sun (high up in the atmosphere) would be the warmest. However, the opposite is true: the atmosphere is primarily heated from the bottom up.
This happens because of the way the atmosphere interacts with different types of radiation.
1. The Atmosphere is “Transparent” to the Sun

2. The Earth is an “Absorber”
The Sun is extremely hot and emits shortwave radiation (mostly visible light).
- Nitrogen, oxygen, and even carbon dioxide are largely “transparent” to these shortwaves.
- The sunbeams pass through the air without bumping into many molecules, meaning they don’t transfer much heat to the upper atmosphere on their way down.
When those sunbeams hit the Earth’s surface (dirt, rocks, pavement, and water), the journey changes.
- The ground is an excellent absorber. It soaks up the solar energy and its temperature rises.
- Now, the ground is hot, and it becomes the primary heat source for the air.
3. The Three-Step Heating Process
Once the ground is warm, it transfers that energy to the atmosphere using the methods we’ve discussed:
- Conduction: The very bottom layer of air touches the hot ground. Heat moves directly into those air molecules.
- Radiation (The Greenhouse Effect): The warm ground emits longwave infrared radiation. Unlike the Sun’s shortwaves, the atmosphere (thanks to water vapor and $CO_2$) is very good at absorbing these longwaves. This traps the heat right near the surface.
- Convection: As that bottom layer of air warms up, it becomes less dense and rises like a hot air balloon. This carries the heat from the surface up into the rest of the sky.
4. The Result: The Lapse Rate
Because the “heater” (the ground) is at the bottom, the air temperature generally drops the higher you go. This is known as the Lapse Rate.
- On average, the temperature drops about 6.5°C for every 1,000 meters you climb.
- This is why there can be snow on top of a mountain even if it’s a warm day in the valley below.
The Exception: Temperature Inversions
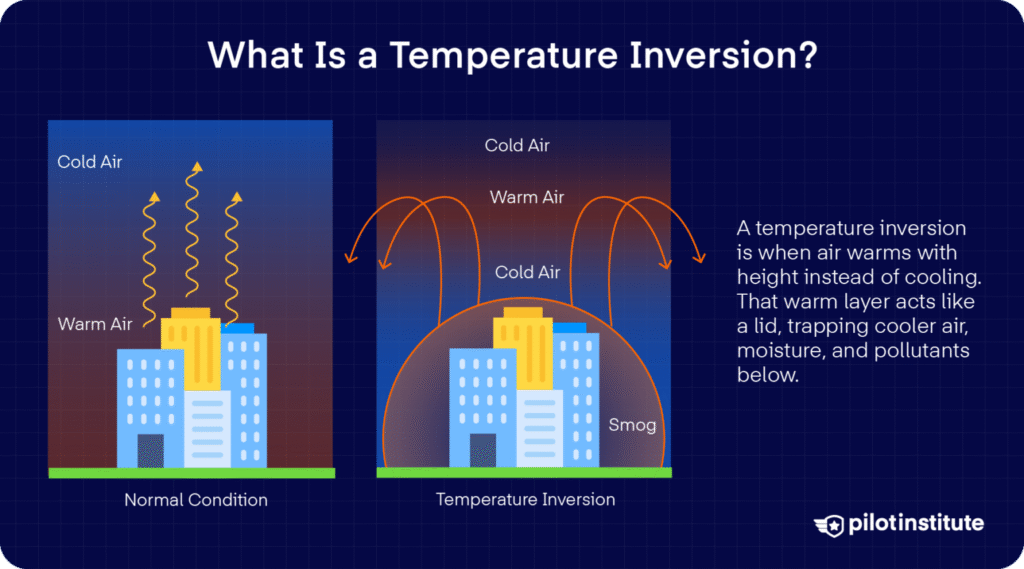
Sometimes, the air doesn’t warm from below. On clear, calm nights, the ground cools off very quickly by radiating its heat into space. The air right above the ground can become colder than the air higher up. This is called a Temperature Inversion, and it can trap smog or fog near the ground because the cold, heavy air can’t rise.
What is the annual energy balance?
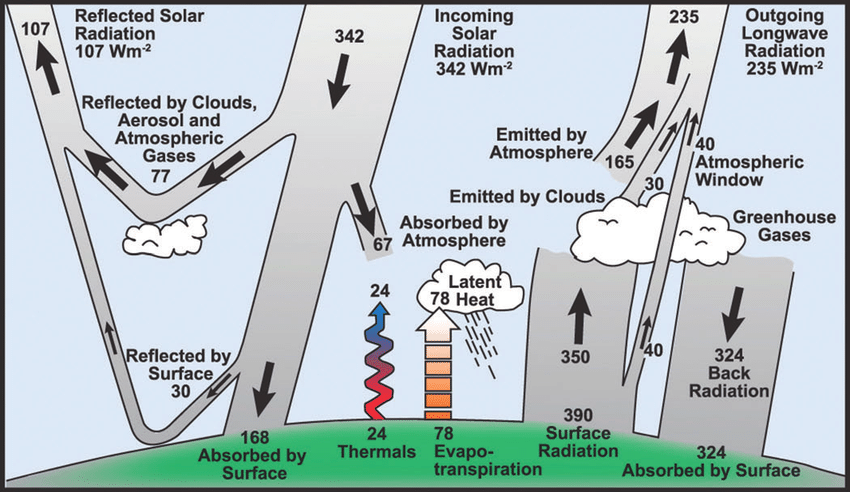
The annual energy balance is the long-term accounting of all solar energy entering the Earth’s system versus the energy leaving it over the course of a full year.
On a global scale, the Earth maintains a nearly perfect balance. If it didn’t—if we kept even 1% more energy than we let out every year—the planet would eventually become a scorched wasteland.
1. The Global “Input-Output” Equation
To calculate the balance, scientists look at the Net Radiation (Q*). For the planet to stay at a stable temperature, the annual sum must be zero:
Q* = Incoming Solar – (Reflected Solar + Outgoing Infrared) = 0
- Income: 100% of the energy comes from the Sun (Shortwave).
- Reflected (Albedo): About 30% is bounced back immediately by clouds, ice, and the atmosphere. This energy never warms the planet.
- Outgoing: The remaining 70% is absorbed, warms the Earth, and is eventually emitted back to space as heat (Longwave).
2. The Latitudinal Imbalance (The “Energy Surplus vs. Deficit”)
While the entire planet is in balance annually, specific parts of the Earth are not. This is one of the most important concepts in meteorology:
- The Tropics (Surplus): Between roughly 35°N and 35°S, the regions receive more solar energy than they radiate back to space. They have an annual energy surplus.
- The Poles (Deficit): Near the North and South Poles, the Earth radiates more heat into space than it receives from the sun. They have an annual energy deficit.
3. Nature’s “Redistribution” System
If the energy stayed where it landed, the equator would get hotter every year and the poles would get colder until they reached absolute zero. To prevent this, the Earth acts like a massive plumbing system:
- Atmospheric Circulation: About 75% of the excess heat is moved toward the poles by winds (the Jet Stream, Trade Winds, and storms).
- Ocean Currents: The remaining 25% is moved by water. Warm currents like the Gulf Stream carry tropical heat thousands of miles north toward Europe.
4. Seasonal Shifts
The balance isn’t steady day-to-day.
- In Summer, a hemisphere has a positive energy balance (it stores heat).
- In Winter, it has a negative energy balance (it loses heat).
- The “Annual” balance is the average of these cycles, ensuring that over 365 days, the Earth returns to its baseline.
5. Why the Balance is Changing
Currently, because of increased greenhouse gases, the Earth’s annual energy balance is slightly positive. We are absorbing about $0.5$ to $1.0$ Watt per square meter more than we are releasing. This small “extra” in the energy bank account is what drives global warming and the melting of polar ice.
What is the daily energy balance?
The daily energy balance (also called the diurnal energy cycle) is the hour-by-hour tracking of how much solar energy a specific location receives versus how much thermal energy it loses.
While the annual balance explains climate, the daily balance explains why the hottest time of day isn’t at noon, and why the coldest time of night is usually just after sunrise.
1. The Daily Cycle: Income vs. Expenses
The daily balance is a competition between Incoming Solar Radiation (shortwave) and Outgoing Terrestrial Radiation (longwave).
- Sunrise to Noon: Incoming solar energy increases as the sun rises higher in the sky.
- Noon: This is the moment of Maximum Solar Intensity. However, it is usually not the hottest time of day.
- Noon to Sunset: The sun begins to set, so incoming energy decreases. However, as long as the incoming solar energy is greater than the heat the Earth is radiating away, the temperature continues to rise.
2. The Afternoon “Thermal Lag”
The highest temperature of the day typically occurs between 2:00 PM and 5:00 PM.
This happens because of Energy Surplus. Even after the sun passes its peak at noon, the “income” of solar energy still outweighs the “expense” of the Earth’s heat loss for several more hours. It’s like a bathtub: even if you turn the faucet down slightly, the water level (temperature) keeps rising as long as the water coming in is faster than the water going down the drain.
3. Nighttime Cooling
Once the sun sets, the “income” drops to zero. However, the “expenses” continue—the Earth continues to radiate infrared heat into space all night long.
- Radiative Cooling: The ground loses heat rapidly. Since the air is warmed from below, the air closest to the ground cools first.
- The Coldest Moment: The lowest temperature usually occurs just after sunrise. Why? Because the Earth has been losing heat all night, and it takes a few minutes of morning sun for the incoming energy to finally overtake the outgoing heat loss.
4. Factors That Influence the Daily Balance
The “shape” of this daily energy curve can be changed by the environment:
- Cloud Cover: Clouds act as a blanket at night, absorbing outgoing radiation and sending it back down, which prevents the temperature from dropping too far. During the day, they act as a shield, reflecting incoming solar energy.
- Humidity: Water vapor is a potent greenhouse gas. Humid areas (like Florida) have very small daily temperature swings because the air holds onto heat. Deserts have massive swings because the dry air allows heat to escape into space instantly at night.
- Surface Type: Asphalt has a high “thermal inertia,” meaning it soaks up energy all day and bleeds it out slowly at night, keeping cities warmer than the surrounding countryside.
Summary Table: A Typical Day
| Time | Energy Status | Temperature Trend |
| Sunrise | Incoming = Outgoing | Daily Minimum |
| Morning | Incoming > Outgoing | Rising |
| Noon | Max Solar Income | Rising |
| Mid-Afternoon | Incoming = Outgoing | Daily Maximum |
| Sunset | Income stops | Falling |
| Overnight | Outgoing only | Falling |
What is the relationship between radiation and energy budgets at the earth’s surface?
The relationship between radiation and energy budgets at the Earth’s surface is essentially an accounting balance. Radiation is the “currency” moving in and out, while the energy budget is the “ledger” that determines if a specific location is getting warmer or colder.
At the surface, this relationship is expressed through the Surface Net Radiation equation.
1. The Net Radiation Equation (Q*)
The energy budget at the surface starts with Net Radiation (Q*), which is the difference between all incoming radiation and all outgoing radiation. It is calculated as:
Q* = (K↓ – K↑) + (L↓ – L↑)
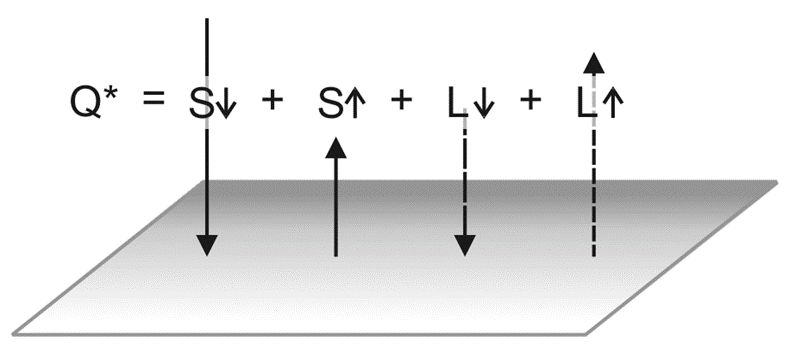
- K (Shortwave/Solar): ↓ is the incoming sunlight; ↑ is the portion reflected back (Albedo).
- L (Longwave/Heat): ↓ is the heat radiated down from the atmosphere; ↑ is the heat emitted by the ground.
2. How Radiation Drives the Budget
Once Q* is calculated, that “Net Energy” doesn’t just sit there. It must be used or moved. The energy budget dictates that the net radiation must be balanced by three main physical processes:
- Sensible Heat (H): This is the energy used to warm the air directly above the surface via conduction and convection. This is the heat you can “sense” with a thermometer.
- Latent Heat (LE): This is the “hidden” energy used for evapotranspiration (evaporating water from soil or plants). This doesn’t raise the temperature; it changes the state of water.
- Ground Heat (G): This is the energy conducted downward into the soil or pavement to be stored during the day.
3. The Relationship in Action
The “budget” changes depending on what the surface looks like. This explains why different places have different climates:
- Over the Ocean: Most of the net radiation goes into Latent Heat (LE). The water evaporates, keeping the air temperature relatively cool and stable.
- In a Desert: There is almost no water for LE. Therefore, nearly all net radiation is forced into Sensible Heat (H), which is why the air temperature sky-rockets during the day.
- In a City: The dark asphalt has a low albedo (absorbs more radiation), and the lack of plants means low LE. The budget shifts heavily toward Sensible Heat and Ground Heat, creating the “Urban Heat Island.”
4. Daytime vs. Nighttime Budgets
The relationship between radiation and the budget flips when the sun goes down:
- Daytime: Q* is usually positive. There is a surplus of energy, so heat moves away from the surface into the air (H) and the ground (G).
- Nighttime: Q* becomes negative. There is no incoming solar radiation, but the Earth continues to emit longwave radiation. The budget must “borrow” energy, so heat flows back from the ground and the air toward the surface to be radiated away into space.
Summary
The Radiation provides the raw energy, but the Energy Budget determines what happens to it. If the surface is wet, the energy becomes moisture in the air; if the surface is dry, the energy becomes heat you can feel.
Solved Problems

Energy Fundamentals
1. Problem: Why does a pot of boiling water stay at 100°C even if you turn the stove to a higher setting? Solution: The extra energy becomes latent heat. Instead of raising the temperature (sensible heat), the energy is used to break the molecular bonds required to change the water from a liquid to a gas.
2. Problem: Why do you feel the heat of a campfire on your face even if the air around you is freezing cold? Solution: This is due to radiation. Unlike conduction or convection, radiation travels through electromagnetic waves and does not need the air to be warm to transfer energy directly to your skin.

3. Problem: Why does a metal spoon in a bowl of hot soup become too hot to touch, while a plastic spoon remains cool? Solution: Metal is an efficient conductor. Its atoms and free electrons easily pass kinetic energy (vibration) from the hot soup up the handle. Plastic is an insulator and resists this transfer of energy.
The Journey of Sunlight
4. Problem: If energy cannot be destroyed, why doesn’t the Earth just keep getting hotter every time the Sun shines? Solution: The Earth maintains a Radiative Equilibrium. It absorbs shortwave radiation from the Sun but emits an equal amount of longwave infrared radiation back into space, keeping the “energy budget” balanced.
5. Problem: Why is it that on a clear day, the sky is blue, but the clouds are white? Solution: It’s a matter of particle size. Tiny gas molecules cause Rayleigh scattering, which redirects blue light. Larger water droplets in clouds cause Mie scattering, which redirects all colors of light equally, blending them into white.
6. Problem: How can a sunbeam take over 100,000 years to reach the Sun’s surface but only 8 minutes to reach Earth? Solution: In the Sun’s dense core, photons undergo a “random walk,” constantly colliding with atoms. Once they reach the surface and enter the vacuum of space, they travel unimpeded at the speed of light.
Atmospheric Mechanics
7. Problem: Why are the bottoms of some clouds flat while the tops are puffy? Solution: This marks the point where convection meets the dew point. Warm air rises until it cools enough for water vapor to release latent heat and condense into liquid droplets, creating a uniform “base” where the phase change begins.
8. Problem: Why is it usually colder on a mountain peak than in the valley, even though the peak is closer to the Sun? Solution: The atmosphere is warmed from below. Sunlight passes through the air and warms the ground; the ground then warms the air via conduction and convection. As you move higher, you move further away from the “heater.”
9. Problem: Why do coastal cities usually have milder temperatures than inland cities at the same latitude? Solution: Water has a high capacity for latent heat and takes longer to warm up or cool down. Oceans act as a “thermal battery,” while dry land converts most solar energy into sensible heat, causing rapid temperature swings.
Daily and Seasonal Cycles
10. Problem: Why is 3:00 PM usually hotter than 12:00 PM, even though the Sun is strongest at noon? Solution: This is the thermal lag. Even after the Sun peaks, the incoming solar energy continues to exceed the Earth’s outgoing infrared radiation for several more hours, causing the temperature to keep climbing.
11. Problem: Why is the coldest time of the night often right after the Sun starts to come up? Solution: The Earth radiates heat all night. It isn’t until the Sun rises high enough for incoming radiation to finally overtake the outgoing loss that the temperature stops falling.
12. Problem: Why do deserts have freezing nights even after blistering hot days? Solution: Deserts lack water vapor (a greenhouse gas). Without moisture to trap outgoing longwave radiation, the heat absorbed during the day escapes almost instantly into space once the Sun sets.
Climate and Surface Energy
13. Problem: How does melting Arctic ice accelerate global warming? Solution: This is an Albedo feedback loop. Ice reflects sunlight (high albedo). When it melts, it reveals dark ocean water which absorbs sunlight (low albedo), warming the planet further and melting more ice.
14. Problem: Why are “Urban Heat Islands” (cities) hotter than the surrounding countryside? Solution: Cities have low latent heat flux (fewer plants/water) and high ground heat flux (asphalt/concrete). Instead of energy being used for evaporation, it is stored in buildings and converted into sensible heat that warms the air.
15. Problem: Why do we have “seasons” if the Sun’s energy output is constant? Solution: It is due to the Angle of Incidence. The Earth’s tilt changes how directly the sunbeams hit the surface. Direct sunlight concentrates energy in a small area, while slanted sunlight spreads that same energy over a larger area, providing less heat.
Atmospheric Energy Quiz
Test your knowledge on radiation laws and energy balance. 10 unique questions with randomized answers appear every time you play.
