Meteorology Today Second Canadian Edition
Earth and Atmospheric Sciences
Think of the relationship between Earth and its atmosphere as a delicate, life-sustaining partnership. They aren’t just sitting next to each other; they are constantly exchanging energy, chemicals, and moisture in a way that keeps our planet from becoming a frozen wasteland or a boiling desert.
Here is a breakdown of how this partnership works:
1. The Earth’s “Security Guard”
The atmosphere acts as a protective shield. Without it, Earth would be vulnerable to the harsh realities of space.
- Radiation Filtering: The ozone layer absorbs most of the sun’s harmful ultraviolet (UV) radiation.
- Meteoric Shield: Most space debris burns up in the mesosphere due to friction before it can hit the surface.
- Temperature Regulation: Through the Greenhouse Effect, the atmosphere traps just enough heat to keep average temperatures around 15°C. Without this, Earth would plummet to roughly -18°C.
2. The Great Exchange
The relationship is a two-way street. The Earth influences the atmosphere just as much as the atmosphere affects the Earth.
The Water Cycle
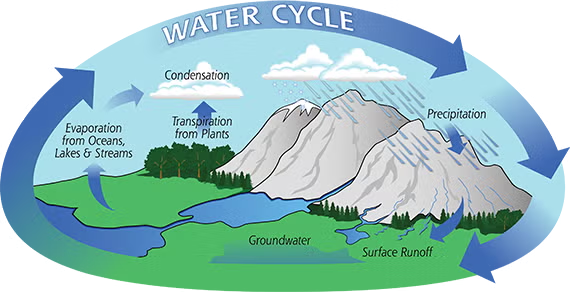
The surface provides the water (oceans, lakes), and the atmosphere provides the transport.
- Evaporation: Heat from the sun turns surface water into vapor.
- Condensation: The atmosphere cools that vapor into clouds.
- Precipitation: The atmosphere returns the water to the surface as rain or snow.
The Carbon-Oxygen Cycle
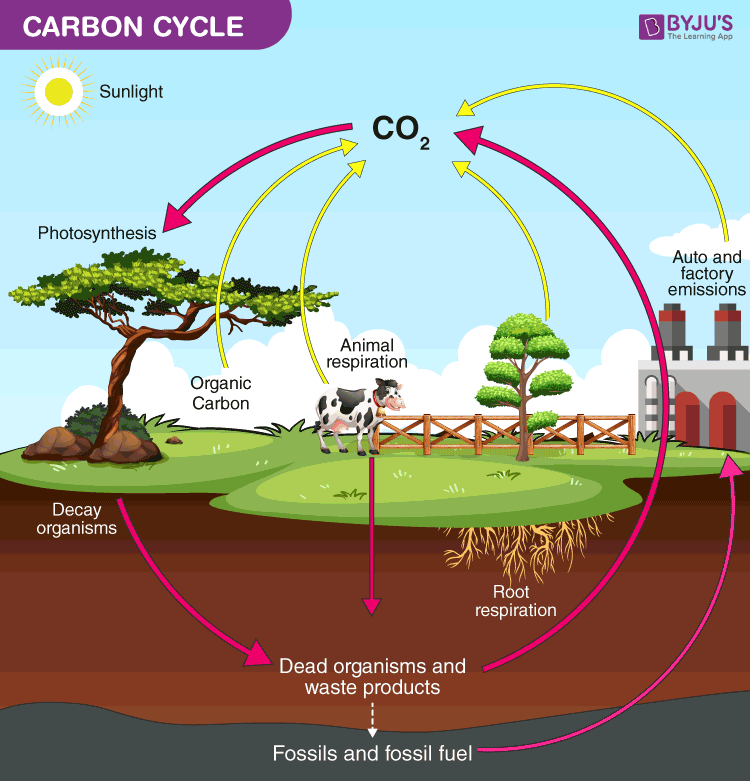
This is the “breathing” part of the relationship.
- Life to Atmosphere: Animals and humans breathe in oxygen and release carbon dioxide (CO2).
- Atmosphere to Life: Plants take in that CO2 and, through photosynthesis, release oxygen back into the air.
3. Gravity: The Glue
The reason we have an atmosphere at all is thanks to Gravity. Earth’s mass creates a gravitational pull strong enough to hold onto its gases, preventing them from drifting off into the vacuum of space. Because gravity is stronger near the surface, the atmosphere is “bottom-heavy”—about 75-80% of the air’s mass is packed into the lowest layer, the Troposphere.
4. Driving the Weather
The uneven heating of the Earth’s surface (the equator gets more sun than the poles) causes the atmosphere to move. This movement creates wind, ocean currents, and weather patterns. In return, these winds help distribute heat around the globe, making more of the planet habitable.
Key Fact: If the Earth didn’t rotate, the relationship would look very different. The Earth’s spin creates the Coriolis Effect, which bends the winds and creates the swirling weather systems we see from space.
What is in a breath of air?
When you take a deep breath, you’re inhaling a complex cocktail of gases. Most people assume it’s mostly oxygen, but in reality, oxygen is actually a minority partner in our air.
Here is the “recipe” for a standard breath of dry air at sea level:
| Component | Percentage | Role / Note |
| Nitrogen (N2) | ~78.08% | The “filler.” It’s mostly inert and dilutes the oxygen so things don’t spontaneously combust. |
| Oxygen (O2) | ~20.95% | The “fuel.” Essential for cellular respiration in humans and animals. |
| Argon (Ar) | ~0.93% | An inert noble gas. It just hangs out and doesn’t react with much of anything. |
| Carbon Dioxide (CO2) | ~0.04% | The “insulator.” Vital for plants and maintaining Earth’s temperature, though levels are rising. |
| Trace Gases | <0.01% | Includes Neon, Helium, Methane, and Krypton. |
The “Invisible” Extras
While the table above covers the dry gases, a real breath of air usually contains a few other key ingredients:
- Water Vapor: Depending on where you are (a desert vs. a rainforest), water vapor can make up anywhere from 0% to 4% of the air.
- Aerosols: These are tiny solid or liquid particles like dust, pollen, sea salt, volcanic ash, or smoke.
- Trace Pollutants: Depending on your environment, you might also be inhaling small amounts of Carbon Monoxide (CO), Nitrogen Dioxide (NO2), or Ozone (O3) at ground level.
What Happens During the Breath?
Your body is remarkably efficient at sorting this mix. When you inhale, your lungs take in that 21% oxygen. However, you don’t use all of it!
When you exhale, the nitrogen remains the same, but the oxygen level drops to about 16%, and the carbon dioxide level jumps from 0.04% to about 4%. Your body essentially swaps a portion of the oxygen for the waste product of your metabolism.
How can the earth’s atmosphere be described?
To describe the Earth’s atmosphere, it’s best to view it not just as “air,” but as a multi-layered heat engine and a biological filter. It is a fluid, dynamic envelope held in place by gravity that becomes thinner and thinner as you move toward space.
Here are the three primary ways scientists describe it:
1. Structural Description: The “Layer Cake”

The atmosphere is divided into distinct layers based on how temperature changes with altitude. We live in the “weather layer” at the bottom, while the outer layers bleed into the vacuum of space.
- Troposphere (0–12 km): Where we live. It contains 80% of the atmosphere’s mass and almost all its water vapor. Temperature decreases as you go higher.
- Stratosphere (12–50 km): Home to the Ozone Layer. Here, temperature actually increases with altitude because the ozone absorbs UV radiation.
- Mesosphere (50–85 km): The coldest layer. This is where most meteors burn up, appearing as “shooting stars.”
- Thermosphere (85–600 km): High-energy X-rays and UV radiation from the sun are absorbed here, making it incredibly hot (though it wouldn’t feel hot to the touch because the air is so thin).
- Exosphere (600 km+): The final frontier where the atmosphere gradually fades into outer space.
2. Chemical Description: The “Homosphere vs. Heterosphere”
We can also describe the atmosphere by how well its gases are mixed.
- The Homosphere (Surface to ~80 km): Throughout this lower section, the chemical composition is remarkably uniform. Whether you are at sea level or on top of Mt. Everest, the ratio of Nitrogen to Oxygen remains roughly 4:1.
- The Heterosphere (Above 80 km): Here, the air is so thin that gases begin to settle out by their atomic weight. Heavier gases (like Nitrogen and Oxygen) stay lower, while lighter gases (like Helium and Hydrogen) float to the top.
3. Functional Description: The “Life Support System”
If the Earth were an apple, the atmosphere would be thinner than the skin. Yet, it performs several critical functions:
- Pressure Maintenance: It provides the atmospheric pressure required for liquid water to exist on the surface. Without it, our oceans would boil away into space.
- The Greenhouse Blanket: It acts as a thermal regulator. It allows short-wave solar energy in but slows down the escape of long-wave heat (infrared) back into space.
- The Chemical Reservoir: It acts as a massive storage tank for the carbon, nitrogen, and oxygen that life requires to cycle through the ecosystem.
Summary Table: Physical Properties
| Property | Value/Description |
| Total Mass | Approximately 5.15 * 1018 kg |
| Surface Pressure | 1013.25 hPa (14.7 psi) at sea level |
| Main Composition | 78% Nitrogen, 21% Oxygen |
| Visual Boundary | The Kármán line (100 km) is the widely accepted “edge of space” |
What is the chemical composition of today’s atmosphere?
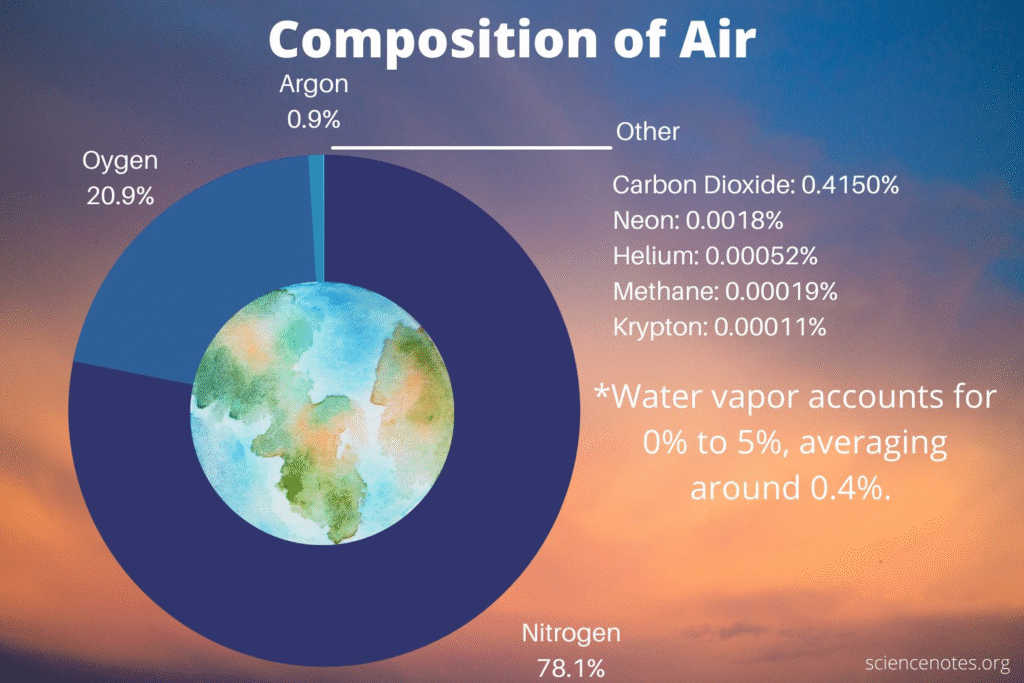
As of February 2026, the atmosphere’s fundamental recipe remains dominated by Nitrogen and Oxygen, but the “trace” ingredients—specifically greenhouse gases—continue to reach record-breaking concentrations.
Here is the chemical breakdown of the air you are breathing today:
1. The Major Constituents (Dry Air)
The bulk of the atmosphere is remarkably stable. Regardless of where you are on Earth, these three gases make up over 99.9% of the dry air.
| Gas | Symbol | Concentration |
| Nitrogen | N2 | 78.08% |
| Oxygen | O2 | 20.95% |
| Argon | Ar | 0.93% |
2. The Variable Components
These are the ingredients that change based on your location and the time of year.
- Water Vapor (H2O): This is the most variable component. In a tropical rainforest, it can make up 4% of the air, while over the Antarctic ice sheet, it may be less than 0.1%.
- Aerosols: Tiny suspended particles like sea salt over the ocean, dust over deserts, or smoke near wildfires.
3. The Trace Gases (2026 Update)
Though they make up less than 0.1% of the atmosphere, these gases dictate our planet’s climate. As of early 2026, several have reached significant new milestones:
Carbon Dioxide (CO2)
- Current Level: Approximately 428 ppm (parts per million).
- Context: For most of human history, this was around 280 ppm. We are currently seeing levels roughly 50% higher than pre-industrial times. In May 2025, a daily record of 431.25 ppm was set at the Mauna Loa Observatory.
Methane (CH4)
- Current Level: Approximately 1,942 ppb (parts per billion).
- Context: Methane levels have increased by about 166% since the year 1750. It is a much more potent heat-trapper than CO2, though it stays in the atmosphere for a shorter time.
Nitrous Oxide (N2O)
- Current Level: Approximately 338 ppb.
- Context: Primarily driven by agricultural fertilizers and industrial processes, this gas is now 25% higher than pre-industrial levels.
Why “Dry Air” Matters
When scientists quote these percentages, they almost always use dry air values. This is because water vapor is so “noisy”—it changes so fast and so often that it would be impossible to give a single number for the whole planet. By “drying” the air in a lab, scientists can get a precise measurement of the other gases to see how human activity is changing the baseline.
What is the nature of the atmospheres of the other planets?
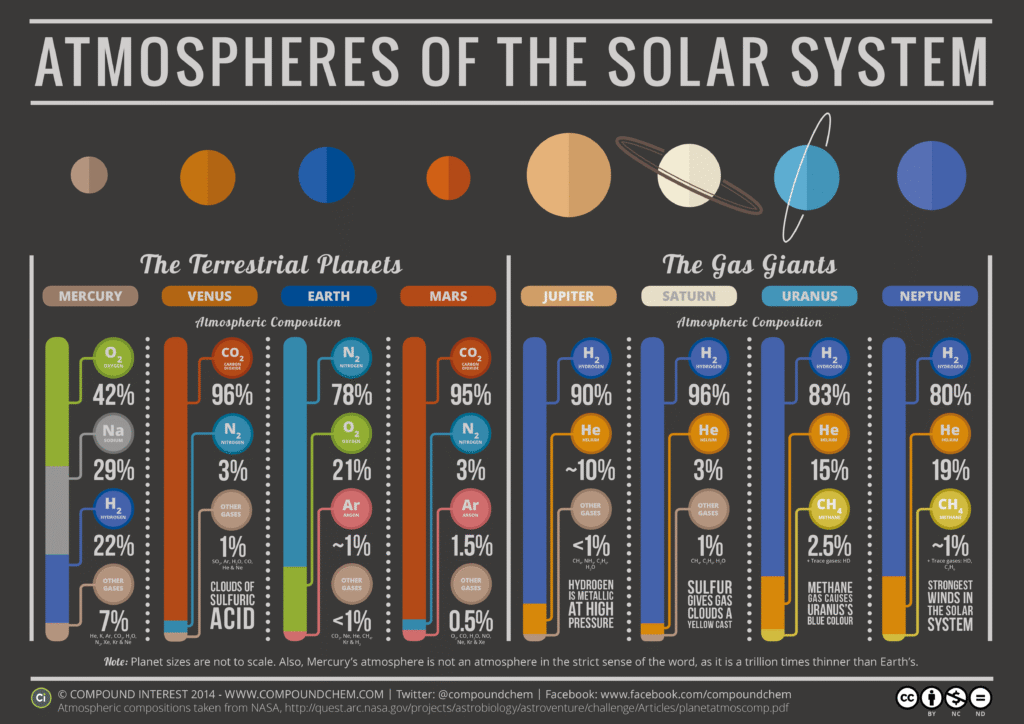
While Earth has a nitrogen-oxygen “security blanket,” the other planets in our solar system have atmospheres that range from thin wisps of gas to crushing, toxic envelopes. In planetary science, the nature of an atmosphere depends on two main things: distance from the sun and the planet’s mass (gravity).
1. The Terrestrial (Rocky) Planets
These planets have “secondary” atmospheres, meaning they were formed by volcanic outgassing or comet impacts rather than being captured from the early solar nebula.
- Mercury: Practically non-existent. It has a thin exosphere of sodium and potassium. Because Mercury is small and close to the sun, solar winds blast its atmosphere away into space.
- Venus: A “runaway greenhouse.” Its atmosphere is 96% Carbon Dioxide and is 90 times thicker than Earth’s. The pressure at the surface is like being 900 meters underwater, and the clouds are made of sulfuric acid.
- Mars: Thin and cold. Also mostly Carbon Dioxide (95%), but it’s 100 times thinner than Earth’s. It’s so thin that liquid water would immediately boil away or freeze.
2. The Gas Giants
Jupiter and Saturn are massive enough to have kept their “primary” atmospheres—the original hydrogen and helium that made up the early solar system.
- Jupiter: Mostly Hydrogen and Helium. It has no solid surface; as you go deeper, the pressure becomes so high that the hydrogen turns into a liquid metal. The colorful bands we see are clouds of ammonia ice and ammonium hydrosulfide.
- Saturn: Similar to Jupiter, but with stronger winds and less distinct cloud bands. It is the least dense planet—if you had a bathtub big enough, Saturn would float.
3. The Ice Giants
Uranus and Neptune are “colder” versions of the gas giants, with a unique chemical signature.
- Uranus & Neptune: While they have hydrogen and helium, they contain a higher proportion of “ices” like water, ammonia, and methane.
- The Blue Hue: Both planets look blue because Methane in their upper atmospheres absorbs red light and reflects blue.
- Winds: Neptune has the fastest recorded winds in the solar system, reaching speeds of over 2,100 km/h.
Comparison at a Glance
| Planet | Major Gases | Surface Pressure | Temperature |
| Venus | CO2, N2 | 92 bar (Extremely High) | 465°C |
| Earth | N2, O2 | 1 bar (Standard) | 15°C |
| Mars | CO2, N2, Ar | 0.006 bar (Very Low) | -60°C |
| Jupiter | H2, He | No solid surface | -110°C (Cloud tops) |
Why is Earth the “Odd One Out”?
Earth is the only planet with a Nitrogen-Oxygen atmosphere. This is entirely due to the presence of life. Early Earth had an atmosphere similar to Mars or Venus (CO2 heavy), but the evolution of photosynthetic bacteria (cyanobacteria) “polluted” the atmosphere with oxygen, permanently changing the planet’s chemistry.
What was the nature of the earth’s early atmosphere?
Earth’s early atmosphere was a hostile, alien environment that would have been instantly lethal to modern life. It has gone through three distinct “versions” over the last 4.5 billion years, evolving from a thin veil of solar gases to the oxygen-rich air we breathe today.
1. The First Atmosphere (The Solar Leftovers)
Immediately after Earth formed, its atmosphere consisted mainly of Hydrogen (H2) and Helium (He). These were the most abundant gases in the early solar nebula.
- The Outcome: This atmosphere didn’t last long. Earth’s gravity was too weak, and its magnetic field hadn’t fully developed. The intense solar wind from the young sun essentially blew these light gases away into space.
2. The Second Atmosphere (The Volcanic Era)
As the Earth’s crust cooled and solidified, intense volcanic activity began to vent gases from the interior—a process called outgassing.
- The Mix: This atmosphere was dominated by Water Vapor (H2O), Carbon Dioxide ($CO_2$), and Nitrogen (N2). There were also trace amounts of methane and ammonia, but zero free oxygen.
- The Great Deluge: As the planet cooled further, the massive amounts of water vapor in the atmosphere condensed and fell as rain for millions of years, creating the first oceans.
- Carbon Scrubbing: Much of the CO2 dissolved into these new oceans and became locked away in carbonate rocks (like limestone), preventing Earth from becoming a “super-hot” version of Venus.
3. The Third Atmosphere (The Biological Revolution)
The most dramatic change in Earth’s history was the Great Oxidation Event about 2.4 billion years ago.
- The Catalyst: Life appeared in the oceans. Specifically, cyanobacteria evolved the ability to perform photosynthesis. They took in CO2 and sunlight and released Oxygen (O2) as a waste product.
- The “Rusting” of Earth: At first, the oxygen didn’t enter the air; it reacted with iron in the oceans, creating massive deposits of iron ore (banded iron formations).
- The Oxygen Spike: Once the minerals on the surface were “saturated,” oxygen began to accumulate in the atmosphere. This killed off most of the existing anaerobic (non-oxygen using) life but paved the way for complex animals and the formation of the Ozone Layer.
Evolution Summary Table
| Feature | Primary Atmosphere | Secondary Atmosphere | Today’s Atmosphere |
| Main Gas | Hydrogen & Helium | Carbon Dioxide & Nitrogen | Nitrogen & Oxygen |
| Oxygen | 0% | 0% | ~21% |
| Source | Solar Nebula | Volcanoes | Life (Photosynthesis) |
| Ability to support life | None | Only anaerobic microbes | Complex multicellular life |
The “Faint Young Sun” Paradox
An interesting mystery of the early atmosphere is that the Sun was about 30% dimmer 4 billion years ago than it is today. Technically, Earth should have been a frozen ball of ice. Scientists believe the extremely high levels of CO2 and Methane in the second atmosphere created a powerful greenhouse effect that kept the planet warm enough for liquid water and life to begin.
What is the atmosphere’s vertical structure?
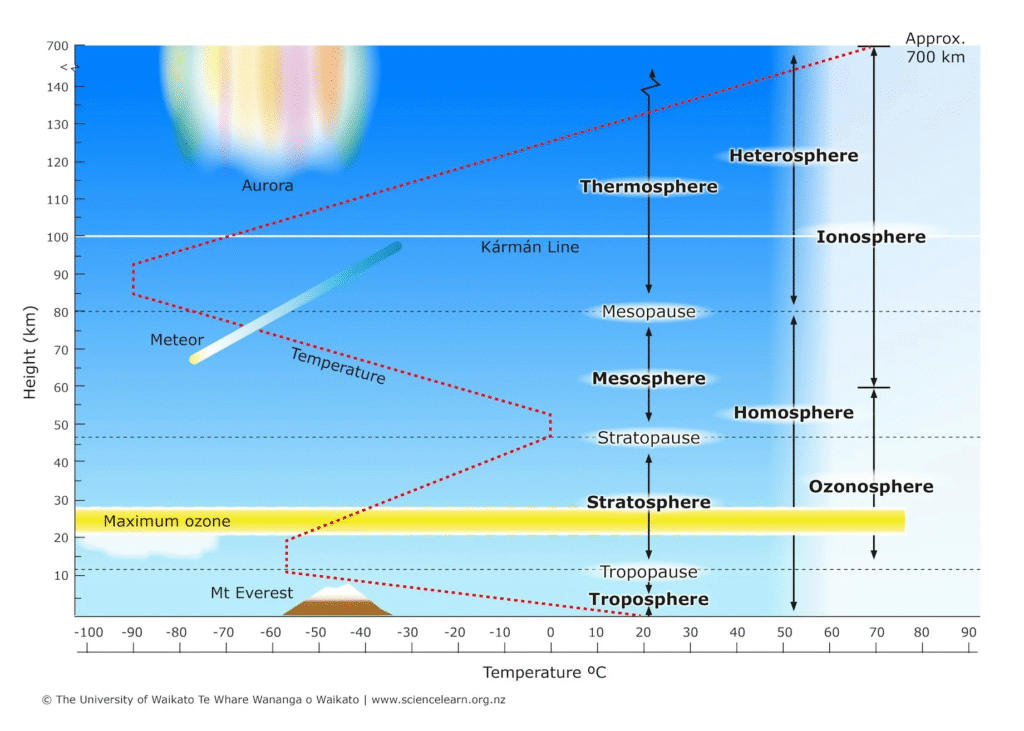
The atmosphere isn’t a uniform “blanket” of air; it is structured like a multi-story building where each floor has its own temperature, density, and chemical behavior. These layers are defined by the Temperature Gradient—whether the air gets hotter or colder as you go up.
1. The Troposphere (0 – 12 km)
This is the “Weather Layer” where we live and breathe.
- Temperature Trend: It gets colder as you go up (about 6.5°C per kilometer). This is because the Earth’s surface, not the sun, is the primary heat source for this layer.
- Key Feature: It contains 99% of the atmosphere’s water vapor and 75-80% of its total mass. The boundary at the top is called the Tropopause, which acts as a “lid” that keeps most weather trapped below.
2. The Stratosphere (12 – 50 km)
This layer is the “Protective Shield.”
- Temperature Trend: It gets warmer as you go up.
- Key Feature: This temperature inversion is caused by the Ozone Layer. Ozone molecules ($O_3$) absorb ultraviolet (UV) radiation from the sun, converting it into heat. Most commercial jets fly in the lower stratosphere to stay above the turbulent weather of the troposphere.
3. The Mesosphere (50 – 85 km)
The “Security Guard” and the coldest part of the atmosphere.
- Temperature Trend: It gets colder again as you go up, reaching temperatures as low as -90°C.
- Key Feature: This is where most meteors burn up. Even though the air is thin, there is enough friction to incinerate space debris before it hits the ground.
4. The Thermosphere (85 – 600 km)
The “Heat Engine.”
- Temperature Trend: It gets extremely hot, with temperatures reaching 1,500°C or more.
- The Catch: If you were there, you would feel freezing cold! The air is so thin (almost a vacuum) that there aren’t enough gas molecules to transfer heat to your skin.
- Key Feature: This is where the International Space Station (ISS) orbits and where the Auroras (Northern and Southern Lights) occur.
5. The Exosphere (600 km+)
The “Exit Ramp.”
- Description: This is the outermost fringe of our atmosphere. The molecules here are so far apart they can travel hundreds of kilometers without hitting each other.
- The End: There is no “hard line” where the atmosphere ends; it simply fades into the vacuum of space.
Summary Table: Vertical Structure
| Layer | Altitude | Temperature Trend | Notable Content |
| Troposphere | 0–12 km | Decreasing | Clouds, storms, life |
| Stratosphere | 12–50 km | Increasing | Ozone layer, jet streams |
| Mesosphere | 50–85 km | Decreasing | Meteor friction, noctilucent clouds |
| Thermosphere | 85–600 km | Increasing | ISS, Auroras, Ionosphere |
| Exosphere | 600 km+ | Variable | Satellites, atoms escaping to space |
Is there a relationship between air density and pressure?
Yes, there is a direct and critical relationship between air density and pressure: they are proportional. If you increase the pressure of a gas (without changing its temperature), you increase its density.
In the atmosphere, both of these properties are governed by gravity and the weight of the air above you.
1. The “Column of Air” Concept
Think of the atmosphere as a giant stack of blankets.
- Pressure is the weight of all those blankets pressing down on you.
- Density is how tightly the fibers of those blankets are packed together.
At sea level, you are at the bottom of the stack. The weight of the entire atmosphere is pressing down, which creates high pressure. Because the air is being squeezed so hard by that weight, the molecules are forced close together, creating high density.
2. The Mathematical Relationship
For those who like the physics of it, the relationship is defined by the Ideal Gas Law. In its simplest form for atmospheric science, it looks like this:
P = ρRT
- P is Pressure
- ρ (rho) is Density
- R is the gas constant
- T is Temperature
If temperature (T) remains constant, as pressure ($P$) goes up, density ($\rho$) must also go up. They move in lockstep.
3. Altitude: The Great Thinner
As you climb a mountain or fly in a plane, both pressure and density drop rapidly, but not at a constant rate. They drop exponentially.
- At 5,500 meters (about 18,000 feet): You are above 50% of the atmosphere’s mass. The pressure is half of what it is at sea level, and the air is only half as dense.
- The “Thin Air” Feeling: When athletes train at high altitudes, they struggle because the air is less dense. While the percentage of oxygen is still 21%, there are physically fewer molecules in every lungful of air you take.

4. The Role of Temperature
Temperature is the “wild card” that can complicate this relationship.
- Cold Air: Molecules move slowly and huddle together. Cold air is more dense and generally creates higher pressure at the surface.
- Warm Air: Molecules bounce around energetically and spread out. Warm air is less dense (which is why hot air balloons rise).
| Situation | Pressure | Density | Molecule Behavior |
| Sea Level | High | High | Squeezed tightly together |
| Mount Everest | Low | Low | Spread far apart |
| Cold Day | Higher | Higher | Clustered together |
| Hot Day | Lower | Lower | Bouncing apart |
What are the atmospheric layers?
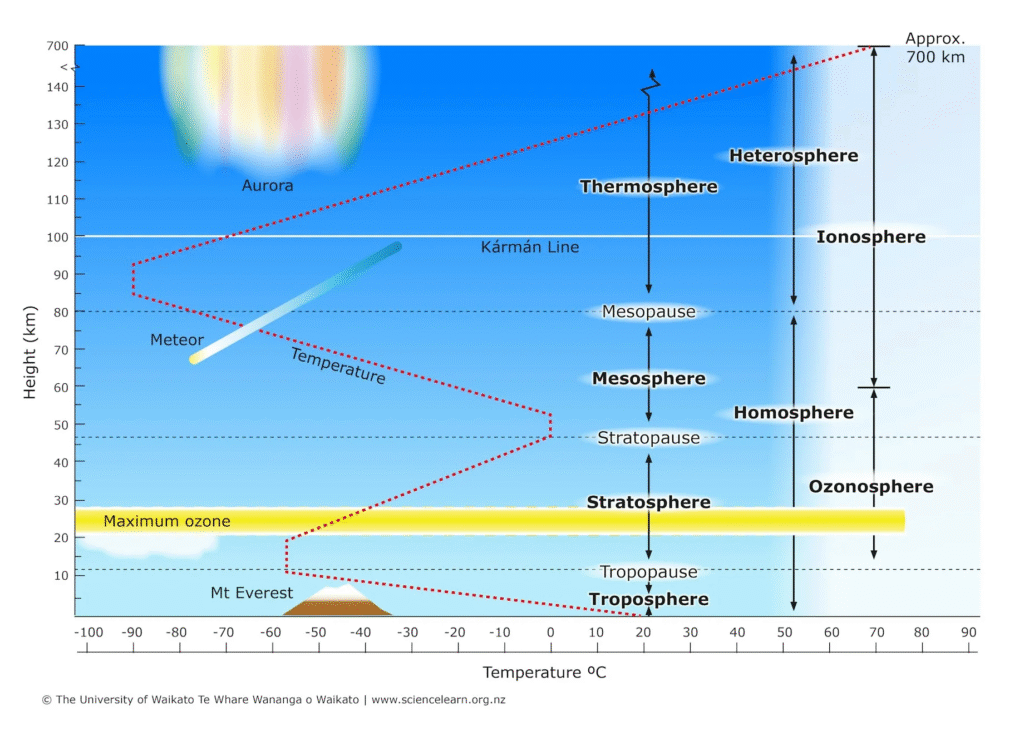
The atmosphere is structured like a multi-story building where each “floor” has its own unique temperature and chemical behavior. These layers are defined by the Temperature Gradient—whether the air gets hotter or colder as you move upward.
1. The Troposphere (0 – 12 km)
This is the “Weather Layer” where we live and breathe.
- Temperature: It gets colder as you go up (about 6.5°C per kilometer). This is because the Earth’s surface, warmed by the sun, is the primary heat source for this layer.
- Key Feature: It contains 99% of the atmosphere’s water vapor and 80% of its total mass. The boundary at the top is called the Tropopause, which acts as a “lid” that keeps most weather trapped below.
2. The Stratosphere (12 – 50 km)
The “Protective Shield.”
- Temperature: It gets warmer as you go up.
- Key Feature: This temperature inversion is caused by the Ozone Layer. Ozone molecules (O3) absorb ultraviolet (UV) radiation from the sun, converting it into heat. Most commercial jets fly in the lower stratosphere to stay above the turbulent weather of the troposphere.
3. The Mesosphere (50 – 85 km)
The “Security Guard” and the coldest part of the atmosphere.
- Temperature: It gets colder again as you go up, reaching temperatures as low as -90°C.
- Key Feature: This is where most meteors burn up. Even though the air is thin, there is enough friction to incinerate space debris before it can hit the ground.
4. The Thermosphere (85 – 600 km)
The “Heat Engine.”

- Temperature: It gets extremely hot, reaching temperatures of 1,500°C or more.
- The Catch: If you were there, you would feel freezing cold! The air is so thin (almost a vacuum) that there aren’t enough gas molecules to bump into your skin and transfer heat.
- Key Feature: This is where the International Space Station (ISS) orbits and where the Auroras (Northern and Southern Lights) occur.
5. The Exosphere (600 km+)
The “Exit Ramp.”
- Description: The outermost fringe. Molecules here are so far apart they can travel hundreds of kilometers without hitting each other.
- The End: There is no “hard line” where the atmosphere ends; it simply fades into the vacuum of space. This is where most geostationary satellites orbit.
Quick Comparison Table
| Layer | Temperature Trend | Key Resident |
| Troposphere | Cooling | Clouds, Storms, Humans |
| Stratosphere | Warming | Ozone Layer, High-altitude Jets |
| Mesosphere | Cooling | Meteors (Burning up) |
| Thermosphere | Warming | ISS, Auroras, Ionosphere |
| Exosphere | Variable | Satellites, Escaping Atoms |
What are solar particles and how do they cause the aurora?

Solar particles are essentially the “exhaust” of the Sun. They consist of a stream of energized, subatomic particles—primarily protons and electrons—that are constantly being blown away from the Sun’s upper atmosphere (the corona) at speeds of up to 900 km/s. This flow is known as the Solar Wind.
When the Sun is particularly active, it releases massive bursts of these particles through events like solar flares or coronal mass ejections (CMEs).
How They Create the Aurora
The Aurora (the Northern and Southern Lights) is the visual result of a collision between these solar particles and Earth’s protective systems. You can think of it as a giant, planetary-scale neon sign.
1. The Magnetic Deflection

As solar particles reach Earth, they encounter our magnetosphere (the magnetic field generated by Earth’s core). Most of these particles are deflected around the planet like water flowing around the bow of a ship.
2. The Weak Points
Earth’s magnetic field lines curve inward and meet the planet at the North and South Poles. Some solar particles get trapped in the magnetic field and are funneled down toward these polar regions.
3. The Collision (The “Glow”)
As these high-energy electrons dive into the upper atmosphere (the thermosphere), they crash into gas molecules like oxygen and nitrogen.
- This collision transfers energy to the gas atoms, “exciting” them.
- To return to their normal state, the atoms must release that extra energy.
- They release it in the form of photons—tiny bursts of light.
Why the Different Colors?
The colors you see in the sky depend on which gas is being hit and how high up the collision occurs:
| Color | Gas Involved | Altitude |
| Pale Green | Oxygen | Most common; occurs up to ~150 km. |
| Red | Oxygen | Rare; occurs at very high altitudes (>250 km). |
| Blue/Purple | Nitrogen | Occurs at lower altitudes; harder for the eye to see. |
Why are they more active right now?
In 2025 and 2026, we are near the Solar Maximum—the peak of the Sun’s 11-year activity cycle. This means the Sun is releasing more CMEs and solar flares than usual, resulting in more frequent and intense auroras that can sometimes be seen much further away from the poles than normal.
Solved Problems

To understand the mechanics of our atmosphere and climate, we can use “back-of-the-envelope” math. These problems focus on the relationships between emissions, temperature, and physical changes using basic arithmetic.
Emissions and Carbon Math
1. The Individual Footprint
If an average person in a developed country generates 15 tons of CO2 per year, and a single mature tree absorbs about 22 kg of CO2 annually, how many trees are needed to offset that one person?

- Solution: Convert tons to kg (15,000 kg / 22 kg ≈ 682 trees).
2. The Daily Global Output
If the world emits roughly 37 billion tons of CO2 per year, how much is emitted every single day?
- Solution: 37,000,000,000 / 365 ≈ 101.4 million tons per day.
3. Methane’s Potency
Methane (CH4) is roughly 28 times more potent at trapping heat than CO2 over a 100-year period. If a farm leaks 10 tons of methane, how many tons of CO2 is that equivalent to (CO2e)?
- Solution: 10 * 28 = 280 tons of CO2e.
4. Parts Per Million (PPM)
If the concentration of CO2 is 428 ppm, what percentage of the atmosphere does CO2 occupy?
- Solution: 428 / 1,000,000 = 0.000428, or 0.0428%.
5. The Coal vs. Gas Divide
Burning coal produces about 95 kg of CO2 per million BTU, while natural gas produces about 53 kg. What is the percentage reduction in emissions if a power plant switches from coal to gas?
- Solution: (95 – 53) / 95 ≈ 44.2% reduction.
Temperature and Energy
6. Celsius to Fahrenheit Trends
Global warming targets are often set at 1.5°C. How many degrees Fahrenheit is a change of 1.5°C? (Note: We are measuring a change in interval, so 1°C change = 1.8°F change).
- Solution: 1.5 * 1.8 = 2.7°F.
7. Solar Constant
The Sun hits the top of our atmosphere with roughly 1,361 Watts per square meter (W/m2). If Earth’s Albedo (reflectivity) is 30%, how much energy is actually absorbed?
- Solution: 1,361 * 0.70 = 952.7 W/m2.
8. Ocean Heat Absorption
The oceans absorb about 90% of the excess heat trapped by greenhouse gases. If the Earth system gains 10 Zettajoules of energy in a year, how much goes into the land and atmosphere combined?
- Solution: 10 ZJ * 0.10 = 1 Zettajoule.
Ice and Sea Level Rise
9. The Melting Point
An ice sheet reflects 80% of sunlight. If it melts and reveals dark ocean water that reflects only 10%, how much more solar energy (as a percentage) is the surface now absorbing?
- Solution: Original absorption = 20%; New absorption = 90%. That is a 350% increase in absorbed energy (70 / 20 = 3.5).
10. Sea Level Math
Thermal expansion and melting ice cause sea levels to rise roughly 3.5 mm per year. How many centimeters will the sea rise in a century at this current rate?
- Solution: 3.5 mm * 100 = 350 mm = 35 cm.
11. Glacier Volume
If a mountain glacier is 2,000 metres long and retreats by 15 meters every year, how many years until the glacier is gone?
- Solution: 2,000 metres / 15 metres ≈ 133.3 years.
Solutions and Efficiency
12. Electric Vehicle (EV) Savings
A gas car gets 30 miles per gallon (mpg). A gallon of gas produces 8.9 kg of CO2. If you drive 12,000 miles a year, how much CO2 do you save by switching to an EV (assuming 0 tailpipe emissions)?
- Solution: (12,000 / 30) * 8.9 = 3,560 kg (3.56 tons) of CO2.
13. Solar Panel Payback
A solar array costs $15,000 to install and saves a homeowner $1,200 a year on electricity. How many years is the “payback period”?
- Solution: $15,000 / $1,200 = 12.5 years.
14. LED Efficiency
An old incandescent bulb uses 60 Watts. An equivalent LED uses 10 Watts. If the lights are on for 5 hours a day, how many Watt-hours are saved daily?
- Solution: (60 – 10) * 5 = 250 Watt-hours.
15. The “Food Mile”
Shipping 1 kg of avocado by plane emits 12 kg of CO2. Shipping it by boat emits 0.2 kg. How many more avocados could you ship by boat for the same “carbon cost” as one air-shipped avocado?
- Solution: 12 / 0.2 = 60 avocados.
The Big Picture
16. Urban Heat Island
An urban area is 4°C warmer than the surrounding countryside. If the countryside is at 28°C, what is the temperature in the city in Fahrenheit?
- Solution: City is 32°C. (32 * 9/5) + 32 = 89.6°F.
17. Deforestation Rate
If the world loses 10 million hectares of forest per year, how many hectares are lost every minute?
- Solution: 10,000,000 / (365 * 24 * 60) ≈ 19 hectares per minute.
18. Renewable Growth
If wind energy capacity is 800 Gigawatts and grows by 10% each year, what will the capacity be in two years?
- Solution: 800 * 1.10 * 1.10 = 968 Gigawatts.
Atmospheric Challenge
10 random questions. Randomized answers. Automatic progression.
