Earth and Atmospheric Sciences
Thermal physics is the branch of science that explores the nature of heat, temperature, and how energy moves through matter.1 It serves as a bridge between the tiny, invisible world of atoms and the large-scale world we experience every day.
At its core, thermal physics asks: Why does heat flow? How do we turn heat into power?2 And what happens to atoms when things get cold?
The Three Pillars of Thermal Physics
Thermal physics isn’t just one subject; it is the integration of three distinct but overlapping fields:3
- Thermodynamics: Focuses on macroscopic (large-scale) properties like pressure, volume, and temperature.4 It provides the “rules of the game” for how energy can be converted from one form to another.5
- Statistical Mechanics: Explains why thermodynamics works by looking at the microscopic level.6 It uses probability and statistics to show how the random motions of trillions of atoms result in the stable temperatures and pressures we measure.
- Kinetic Theory: Specifically deals with gases. It treats gas particles as tiny billiard balls constantly colliding with each other and the walls of their container to explain concepts like pressure.
The Four Laws of Thermodynamics
These laws are the fundamental “commandments” of the universe regarding energy:7
- Zeroth Law (Thermal Equilibrium): If two systems are both in equilibrium with a third system, they are in equilibrium with each other.8 This is why thermometers work.
- First Law (Conservation of Energy):9 Energy cannot be created or destroyed, only transformed.10 The change in internal energy (dU) is equal to the heat added to the system (dQ) minus the work done by the system (dW): dU = dQ – dW
- Second Law (Entropy): In any energy transfer, some energy is always wasted.15 The total “disorder” (entropy) of the universe always increases.16 Heat will never spontaneously flow from a cold object to a hot one.17
- Third Law (Absolute Zero):18 As a system approaches absolute zero (19 0 K or 20 -273.15°C), its entropy reaches a minimum constant value.21 In practice, it is impossible to reach absolute zero.22
How Heat Moves
Heat is simply the transfer of thermal energy between substances.23 It happens in three ways:24
Explore
- Conduction: Heat transfer through direct contact (e.g., a metal spoon getting hot in a cup of coffee).25
- Convection: Heat transfer through the movement of fluids like air or water (e.g., warm air rising above a radiator).26
- Radiation: Heat transfer via electromagnetic waves (e.g., feeling the warmth of the Sun through the vacuum of space).27
Why It Matters
Thermal physics is responsible for almost every convenience of modern life:
- Engines & Power: From car engines to nuclear power plants, these systems turn heat into mechanical or electrical energy.28
- Climate Science: Understanding how the atmosphere traps heat (the greenhouse effect) is entirely based on thermal physics.
- Daily Tech: Your smartphone needs thermal management to keep its processor from melting, and your refrigerator uses “phase changes” to pump heat out of your food.
Would you like me to explain a specific concept further, such as how entropy relates to the “arrow of time”?
Thermodynamics in everyday life
This video provides practical examples of how the principles of heat and energy conversion apply to common activities like cooking and home heating.
While people often use the words interchangeably, in physics, temperature and heat describe two very different concepts.1
The simplest way to remember the difference is: Temperature is a “status,” while heat is a “transfer.”2
1. Temperature: The Average “Vibe”
Temperature is a measure of the average kinetic energy of the particles in a substance.3
- What it tells you: How fast the atoms or molecules are moving or vibrating on average.4
- Measurement: It is measured in degrees (Celsius, Fahrenheit) or Kelvin using a thermometer.5
- Key Characteristic: It is “intensive,” meaning it doesn’t depend on how much of the substance you have.6 A cup of boiling water and a giant pot of boiling water are both exactly 100°C.
2. Heat: The Flow of Energy
Heat is the total energy that moves from one object to another because of a temperature difference.7
- What it tells you: How much thermal energy is being transferred. An object doesn’t “have” heat; heat is what happens when energy is on the move.
- Measurement: It is measured in Joules (J) or Calories using a calorimeter.8
- Key Characteristic: It is “extensive,” meaning it does depend on the amount of substance.
Comparison Table
| Feature | Temperature (T) | Heat (Q) |
| Definition | Average kinetic energy of particles | Total energy transferred between objects |
| Unit | Celsius (°C), Kelvin (K) | Joules (J), Calories (cal) |
| Tool | Thermometer | Calorimeter |
| Depends on Mass? | No (Intensive) | Yes (Extensive) |
| Analogy | The speed of individual cars | The total traffic passing through a gate |
The “Iceberg vs. Coffee” Example
This classic example helps visualize the difference:
- A cup of hot coffee has a higher temperature than an iceberg. Its molecules are moving much faster.9
- An iceberg has much more thermal energy (and potential for heat transfer) than the coffee. Because it is so massive, the total energy of all its trillions of slow-moving molecules far exceeds the energy in the small cup of coffee.
If you poured that coffee onto the iceberg, heat would flow from the coffee to the ice because of the temperature difference, even though the iceberg has more total energy.10
Would you like to see how we calculate exactly how much heat is needed to raise an object’s temperature using the Q = mcΔT formula?
Difference between heat and temperature
This video offers a great visual breakdown of why these two concepts are different and how they interact in everyday physics experiments.
What are the common temperature scales?
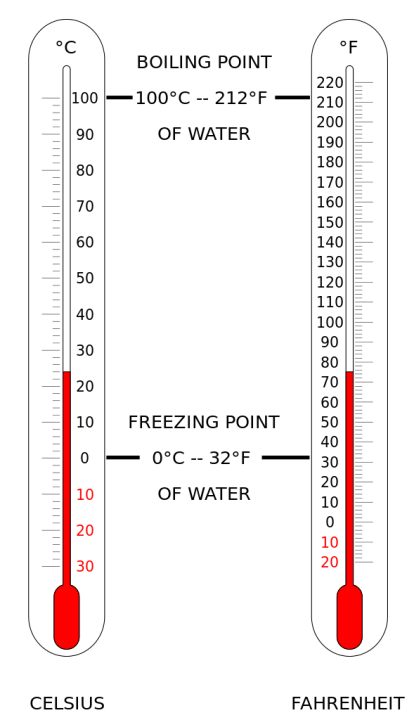
There are three primary temperature scales used worldwide: Celsius, Fahrenheit, and Kelvin.1 Each was designed with a different starting point and purpose in mind.2
1. Celsius (3 °C)4
Also known as the centigrade scale, this is the most common scale used globally for weather and daily life.5
- Reference Points: Based on water.6 It sets 7 0°C as the freezing point and 8 100°C as the boiling point.9
- Usage: Used in almost every country outside the United States and for most general scientific applications.10
2. Fahrenheit (°F)
Primarily used in the United States and a few Caribbean countries.11
- Reference Points: Water freezes at 12 32°F and boils at 13 212°F.14
- History: It was originally designed around human comfort and brine (saltwater) freezing points.15 A “degree” in Fahrenheit is smaller than a “degree” in Celsius.16
3. Kelvin (17 K)18
The Kelvin scale is the “absolute” scale used in physics and chemistry.19
- Reference Points: It begins at absolute zero (20 0 K), the theoretical point where all molecular motion stops.21 There are no negative numbers in Kelvin.22
- Relationship: One Kelvin is the same “size” as one degree Celsius.23 0°C is equal to 24 273.15 K.25
- Note: We do not use the word “degrees” with Kelvin (e.g., it’s “300 Kelvin,” not “300 degrees Kelvin”).26
Comparison Table
| Event | Fahrenheit (∘F) | Celsius (∘C) | Kelvin (K) |
| Absolute Zero | -459.67 | -273.15 | 0 |
| Water Freezes | 32 | 0 | 273.15 |
| Room Temp | 68 | 20 | 293.15 |
| Body Temp | 98.6 | 37 | 310.15 |
| Water Boils27 | 212 | 100 | 373.15 |
Conversion Formulas
If you need to switch between them, here are the standard mathematical relationships:
- Celsius to Fahrenheit: 28 F = 9/5C + 32
- Fahrenheit to Celsius: 29 C = 5/9(F – 32)
- Celsius to Kelvin: 30 K = C + 273.15
Would you like me to calculate a specific temperature conversion for you?
Temperature Scales and how they are used
This video explores the history behind these scales and explains why the world uses different systems for weather, comfort, and science.
What is the Kelvin temperature scale?
The Kelvin (K) scale is the primary system for measuring temperature in the physical sciences.1 It is an “absolute” scale, meaning it is not based on the properties of a specific substance (like the freezing point of water) but on the fundamental laws of physics.2
What Makes Kelvin Different?
Unlike Celsius and Fahrenheit, the Kelvin scale starts at Absolute Zero (3 0 K).4
- Absolute Zero: This is the theoretical point where all molecular motion stops.5 You cannot have a temperature lower than 6 0 K, which means there are no negative numbers on this scale.7
- The Increment: One Kelvin is exactly the same “size” as one degree Celsius.8 The only difference is where they start counting.
- Terminology: You do not use the word “degree” or the symbol (9 °) with Kelvin.10 You simply say “300 Kelvin” or “300 K.”11
Why Scientists Use It
Kelvin is the “go-to” scale for thermodynamics and chemistry for several practical reasons:12
- Direct Proportionality: In scientific equations (like the Ideal Gas Law), temperature must represent the actual amount of energy in a system.13 If you double the Kelvin temperature (e.g., from 200 K to 400 K), you have exactly doubled the average kinetic energy of the particles. This is not true for Celsius.
- No Math Errors: Because there are no negative numbers, you avoid the mathematical impossibility of having “negative pressure” or “negative volume” in equations where temperature is a multiplier.14
- Universal Standards: It is the official International System of Units (SI) unit for temperature, ensuring scientists in every country use the same baseline for their data.15
Kelvin vs. Celsius Benchmarks
Since the two scales use the same increment, converting is easy: just add 273.15 to your Celsius temperature.
| Milestone | Celsius (∘C) | Kelvin (K) |
| Absolute Zero | -273.15°C | 0 K |
| Freezing point of water | 0°C | 273.15 K |
| Room Temperature | 20°C | 293.1 K |
| Boiling point of water | 100°C | 373.15 K |
Why the Kelvin Scale is considered a thermodynamic scale
This video explains why starting at absolute zero makes the Kelvin scale essential for calculating energy changes in thermodynamics.
What are thermometers?
Thermometers are scientific instruments used to measure temperature or temperature gradients.1 Every thermometer works on a single fundamental principle: they contain a “sensing element” that changes in a predictable way when it gets hot or cold.2
A thermometer consists of two essential parts:3
- A sensor: Something that reacts to temperature (like a liquid that expands or a metal that changes electrical resistance).4
- A scale: A way to convert that physical change into a numerical value (like the markings on a glass tube or a digital screen).5
How Different Thermometers Work
Different types of thermometers use different physical properties to “feel” the temperature.6
1. Liquid-in-Glass (The Classic)7
This is the traditional thermometer containing mercury or colored alcohol.8
- The Science: It relies on thermal expansion.9 When the liquid gets hot, its molecules move faster and take up more space, forcing the liquid up the narrow tube.10
- Usage: Used in labs and spirit-filled outdoor weather thermometers. Mercury versions are being phased out due to toxicity.11
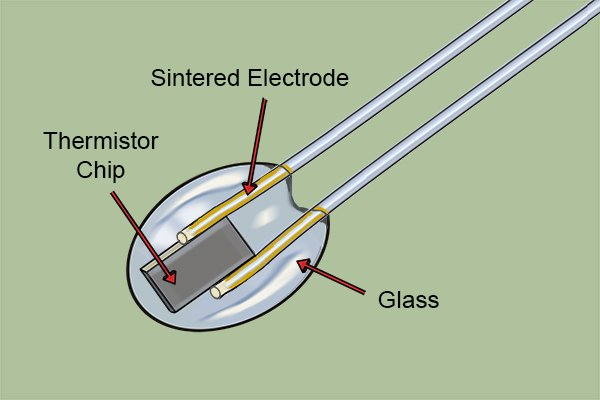
2. Digital (Electronic)
These are the most common thermometers found in homes today.
- The Science: They use a thermistor—a type of resistor whose electrical resistance changes significantly with temperature.12 A small computer chip measures this resistance and converts it into a digital number.13
- Usage: Fever thermometers, digital kitchen probes, and indoor digital displays.
3. Infrared (Non-Contact)14
Commonly used for “no-touch” forehead scans or checking hot oven surfaces.
- The Science: Every object emits invisible infrared radiation.15 The hotter the object, the more radiation it emits.16 The thermometer focuses this light onto a detector (a thermopile) that converts the light energy into a temperature reading.
- Usage: Medical screenings, cooking, and industrial maintenance.17
4. Bimetallic Strip (The Mechanical Dial)18
Often found in older thermostats or analog meat thermometers.
- The Science: It uses two different metals (like steel and copper) bonded together.19 Because different metals expand at different rates, the strip is forced to bend as it heats up.20 This bending motion moves a needle on a dial.
- Usage: Oven thermometers, grill dials, and industrial gauges.
Summary of Common Types
| Type | Sensing Method | Best For… |
| Digital | Electrical resistance | Accuracy and speed in medical/home use |
| Infrared | Infrared radiation | Fast, non-contact surface measurement |
| Liquid-in-Glass | Volume expansion | Simple lab work or weather monitoring |
| Bimetallic21 | Mechanical bending22 | Rugged, battery-free oven or grill monitoring23 |
| Thermocouple | Voltage difference | Industrial extremes (furnaces, engines) |
What is linear thermal expansion?
Linear thermal expansion is the increase in the length of a solid object as its temperature rises.1
While most objects expand in all directions (volume expansion), we focus on “linear” expansion when an object is much longer than it is wide—like a wire, a metal rod, or a structural beam.2
Why It Happens
At the microscopic level, all solids are made of atoms that are constantly vibrating.3 When you add heat, you are giving these atoms more kinetic energy.4
- As they vibrate more violently, they push against their neighbors with more force.5
- This increased movement forces the atoms to maintain a greater average distance from each other.6
- The result is that the entire object “stretches” or grows longer.
The Formula
To calculate how much an object will expand, physicists use a simple linear relationship:
ΔL = αL0ΔT
- ΔL: The change in length (how much it grew).7
- α (alpha): The coefficient of linear expansion.8 This is a constant value unique to each material (e.g., steel expands differently than aluminum).
- L0: The original length of the object.9
- ΔT: The change in temperature.10
Real-World Examples
Engineers must account for this expansion to prevent structures from breaking or warping:11
- Railroad Tracks: You may have seen small gaps between segments of track.12 These gaps give the steel room to expand on hot summer days.13 Without them, the tracks would buckle and warp (a phenomenon called “sun kink”).14
- Bridges: Most bridges have “expansion joints”—those metal, comb-like teeth in the road surface.15 They allow the bridge to get longer in the summer and shorter in the winter without cracking the concrete.16
- Power Lines: You’ll notice that power lines sag more in the summer.17 This is because the wires have expanded in length due to the heat.18
- The Eiffel Tower: Because it is made of iron, the Eiffel Tower can actually grow about 15 cm (6 inches) taller during a hot summer day compared to winter.19
Summary of Expansion Types
While linear expansion is the most commonly studied in introductory physics, it is part of a larger family:
- Linear Expansion (20 α): Change in length.21
- Area Expansion (22 β): Change in surface area (23 β ≈ 2α).24
- Volume Expansion (25 γ): Change in total 3D space (26 γ ≈ 3α).27
Would you like to try a practice calculation using a specific material like steel or copper?
Thermal Expansion of Solids and Liquids
This video provides a deeper dive into the relationship between linear and volume expansion coefficients, helping to visualize how length changes translate into overall size increases.
What is volume thermal expansion?
Volume thermal expansion is the increase in the total three-dimensional space an object occupies as its temperature rises.1 While linear expansion deals only with length, volume expansion accounts for changes in length, width, and height simultaneously.
This phenomenon applies to solids, liquids, and gases.2
The Formula
The change in volume is proportional to the original volume and the change in temperature:3
ΔV = βV0ΔT
- ΔV: The change in volume (how much the object expanded).4
- β (beta): The coefficient of volume expansion.5 This value is specific to each material.
- V0: The original (initial) volume.6
- ΔT: The change in temperature.7
The Relationship to Linear Expansion
For most solid materials that are isotropic (they expand the same in all directions), the volume coefficient is approximately three times the linear coefficient:
β ≈ 3α
Key Differences Across States of Matter
| State | Expansion Level | Notes |
| Solids | Small | Atoms are tightly bound; they vibrate more but stay in fixed positions. |
| Liquids | Moderate | Liquids generally expand more than solids. They have no linear expansion, only volume. |
| Gases | High | Gases expand significantly because their molecules are not bound together. |
Real-World Examples
- Gasoline Overflow: If you fill your car’s gas tank to the brim on a cool morning and then park in the hot sun, the gasoline will expand more than the steel tank.9 This often leads to gasoline dripping out of the overflow valve.
- Liquid Thermometers: When the liquid (mercury or alcohol) inside a thermometer is heated, its volume increases.10 Since it is confined to a narrow tube, the only way it can expand is upward, showing a higher temperature on the scale.
- Ocean Level Rise: One of the primary drivers of rising sea levels is not just melting ice, but the thermal expansion of seawater. As the global ocean temperature rises, the water literally takes up more space.
- The “Anomalous” Case of Water: Water is a rare exception.11 While most substances contract as they cool, water actually expands as it cools from 12 4°C to 13 0°C.14 This is why ice floats and why pipes burst in the winter.
Would you like to see a sample calculation, perhaps comparing how much a tank of gasoline expands compared to the tank itself?
Thermal Expansion of Solids and Liquids
This video provides a deeper dive into the relationship between linear and volume expansion coefficients, helping to visualize how length changes translate into overall size increases.
What is the relationship between heat and internal energy?
In physics, the relationship between heat and internal energy is best understood as the difference between energy in transit and energy in storage.
Internal energy is a property that a system has, while heat is a process by which that energy is changed.1
1. Defining the Two Concepts
- Internal Energy (2 U): This is the total energy stored within a substance.3 It is the sum of all microscopic kinetic energy (atoms moving, rotating, or vibrating) and potential energy (forces between molecules).4 A system “possesses” internal energy just like a bank account possesses a balance.5
- Heat (6 Q): This is the transfer of energy across a system’s boundary due to a temperature difference.7 A system does not possess heat; heat only exists when energy is moving from a warmer object to a cooler one.8
2. The First Law of Thermodynamics
The definitive relationship between the two is found in the First Law of Thermodynamics, which is essentially a law of energy bookkeeping:9
ΔU = Q – W
- ΔU (Change in Internal Energy): The net increase or decrease in the energy stored in the system.10
- Q (Heat): Energy added to the system.11
- W (Work): Energy leaving the system because it did work (like a gas pushing a piston).
The Analogy: If your Internal Energy is the balance in your bank account, Heat is like making a deposit.12 The deposit increases your balance, but once the money is in the account, you just call it “your balance”—you don’t call it “the deposit” anymore.
3. Key Differences at a Glance
| Feature | Internal Energy (U) | Heat (Q) |
| Nature | A state function (describes a system’s current condition). | A path function (describes a process or change). |
| Ownership | A system “has” internal energy. | A system “exchanges” heat. |
| Components | Microscopic kinetic + potential energies. | Energy in transit due to ΔT. |
| At Equilibrium | Remains constant. | Becomes zero (no heat flows between objects at the same T). |
4. How Heat Changes Internal Energy
When heat is transferred into a system, one of two things usually happens to the internal energy:
- Temperature Change: The added energy increases the kinetic energy of the particles, and the temperature rises.13
- Phase Change: The added energy goes into breaking or weakening the bonds between molecules (changing potential energy), causing the substance to melt or boil without a change in temperature.14
The First Law of Thermodynamics and Internal Energy
This video provides a clear visual explanation of how temperature, internal energy, and heat differ, specifically focusing on the microscopic motion of particles.
What is specific heat capacity?
Specific heat capacity (often shortened to “specific heat”) is a physical property that tells us how much energy is needed to change the temperature of a specific amount of a substance.1
It explains why a metal spoon in a cup of hot coffee gets hot almost instantly, while the coffee itself takes much longer to cool down.
The Definition
Specific heat capacity (2 c) is defined as the amount of heat energy required to raise the temperature of one unit of mass (usually 1 kg or 1 g) of a substance by one degree (either 3 1°C or 41 K).5
The Formula
To calculate the total heat energy (Q) absorbed or released by an object, we use this fundamental equation:
Q = mcΔT
- Q: Heat energy (measured in Joules, 6 J)7
- m: Mass of the substance (kg or g)8
- c: Specific heat capacity (9 J/kg • K or 10 J/g • °C)11
- ΔT: Change in temperature (12 Tfinal – Tinitial)13
Common Specific Heat Values
Materials with a high specific heat are resistant to temperature changes (they heat up and cool down slowly).14 Materials with a low specific heat change temperature very quickly.
| Substance | Specific Heat (J/kg⋅∘C) | Characteristics |
| Water | 4,184 | Very High: Great for cooling engines and regulating climate. |
| Wood | 1,700 | Moderate: Why a wooden spoon doesn’t burn your hand. |
| Aluminum | 900 | Low-Moderate: Heats up quickly in a pan. |
| Glass | 840 | Low: Heats up faster than water. |
| Iron/Steel | 450 | Low: Used in radiators because it transfers heat fast. |
| Gold | 129 | Very Low: Changes temperature with very little energy. |
Why Is Water Special?
Water has one of the highest specific heat capacities of any common substance.15 This has massive real-world implications:
- Climate Control: Oceans absorb huge amounts of solar energy during the day without getting much hotter, and release it slowly at night.16 This keeps coastal cities from having extreme temperature swings.
- Body Temperature: Since humans are mostly water, our high specific heat helps us maintain a stable internal temperature even when the weather changes.
- Cooking: This is why a pot of water takes a long time to reach a boil, but once it’s hot, it stays hot for a long time.
Specific Heat vs. Heat Capacity
People often confuse these two, but there is a key difference:
- Specific Heat Capacity (c): An intrinsic property of the material.17 It doesn’t matter if you have a drop of water or an ocean; the specific heat is the same (18 4,184 J/kg°C).19
- Heat Capacity (C): An extrinsic property of the object. It is the total energy needed to heat up the entire thing.20 A bathtub full of water has a much higher heat capacity than a cup of water, even though they have the same specific heat.
This video offers a great visual breakdown of why different substances react differently to the same amount of energy, helping you visualize the microscopic “jiggling” of atoms.
What is the latent heat of phase change?
Latent heat is the energy absorbed or released by a substance during a change in its physical state (phase) that occurs without a change in temperature.1
The word “latent” comes from the Latin word for “hidden.”2 It is called this because, unlike “sensible heat” which you can feel and measure with a thermometer as a temperature rise, latent heat is “hidden” in the breaking or forming of molecular bonds.
Why Doesn’t the Temperature Change?
Normally, adding heat to a substance makes its molecules move faster, which increases the temperature. However, during a phase change:
- The energy goes into the bonds: Instead of making molecules move faster, the energy is used to overcome the attractive forces (bonds) holding the molecules together.3
- The “Flat Line” on a graph: If you graph temperature over time as you heat ice, you will see a flat horizontal line at 4 0°C and 5 100°C.6 The temperature stays exactly the same until every single gram of the substance has finished changing phase.7
The Two Main Types
- Latent Heat of Fusion (8 Lf): The energy required to change a substance from solid to liquid (melting) or released when changing from liquid to solid (freezing).9
- Latent Heat of Vaporization (10 Lv): The energy required to change a substance from liquid to gas (boiling/evaporation) or released when changing from gas to liquid (condensation).11
The Formula
To calculate the total heat (Q) involved in a phase change, we use:
Q = mL
- Q: Total heat energy (Joules, J)
- m: Mass of the substance (kg or g)12
- L: Specific latent heat of the substance (13 J/kg)14
Example: Water
Water requires a massive amount of energy to change phase compared to just heating it up:
- To melt 1 kg of ice: You need 15 334,000 J (16 Lf).17
- To boil 1 kg of water: You need 2,260,000 J (Lv).
Note: It takes nearly seven times more energy to turn boiling water into steam than it does to heat that same water from freezing all the way to boiling!
Real-World Applications
- Steam Burns: Steam at 18 100°C is much more dangerous than boiling water at 19 100°C.20 When steam hits your skin, it condenses into liquid, releasing all that “hidden” latent heat of vaporization directly onto your skin.
- Sweating: When you sweat, the water on your skin evaporates. To do this, it must absorb the latent heat of vaporization from your body, which carries away a huge amount of heat and cools you down.
- Protecting Crops: Farmers sometimes spray water on fruit trees when a freeze is expected. As the water freezes into ice on the branches, it releases latent heat into the tree, often keeping the fruit just above the temperature where it would be damaged.
What does equilibrium between phases of matter mean?
Earth and Atmospheric Sciences
What is humidity?
Humidity is the amount of water vapor present in the air.1 While water vapor is an invisible gas, its presence significantly affects the weather, how we feel (comfort), and various physical processes like evaporation and condensation.2
In thermal physics, humidity is usually discussed in two distinct ways: Absolute and Relative.3
1. Absolute Humidity
Absolute humidity is the total mass of water vapor in a specific volume of air, regardless of the temperature.4
- How it’s measured: Usually in grams of water vapor per cubic meter of air (5 g/m3).6
- The Concept: Think of it as the “literal” amount of water in the air. If you could “wring out” a cubic meter of air like a sponge, the amount of liquid water you’d get is the absolute humidity.
2. Relative Humidity (RH)
This is the measurement you most often hear in weather reports. It is the ratio of the current amount of water vapor in the air compared to the maximum amount the air could hold at its current temperature.7
- How it’s measured: As a percentage (8 0% to 9 100%).10
- The Key Rule: Warm air can hold more water vapor than cold air.11 * If you have a fixed amount of water vapor and you increase the temperature, the relative humidity decreases (because the “container” got bigger).
- If you decrease the temperature, the relative humidity increases.12
Important Concepts Related to Humidity
The Dew Point
The dew point is the temperature at which air becomes 100% saturated (13 100% RH).14 If the temperature drops any further, the air can no longer hold all the water vapor, and the excess must condense into liquid.15
- On the ground: This creates dew.16
- In the air: This creates fog or clouds.17
- On a cold glass: This creates the “sweat” on the outside of your drink.
Evaporative Cooling and Comfort
Humidity affects how we regulate body temperature.18
- Low Humidity: Sweat evaporates quickly, which removes heat from your body (latent heat of vaporization) and cools you down.19
- High Humidity: The air is already “full” of water vapor, so sweat cannot evaporate.20 Instead, it sits on your skin, making you feel “sticky” and much hotter than the actual thermometer reading.21 This is why a 30°C day in a rainforest feels much more oppressive than 30°C in a desert.
Comparison Summary
| Type | Definition | Units | Depends on Temperature? |
| Absolute Humidity | Mass of water per volume of air | g/m3 | No |
| Relative Humidity | Ratio of current vapor to max capacity | % | Yes (inverse relationship) |
| Specific Humidity | Mass of water vapor per mass of air | g/kg | No |
Solved Problems
Learn how we bridge these gaps: [The Starline Philosophy: The Modern Polymath]
