Free Asteroids – Play Game Instantly!
Why is it important to understand the earth system?
Earth and Atmospheric Sciences
To understand the relationship between minerals and rocks, it is helpful to think of them like ingredients in a recipe. In the same way that flour, sugar, and cocoa are the “building blocks” of a chocolate cake, minerals are the individual components that come together to form a rock.
What is a Mineral?
A mineral is a solid, naturally occurring substance that is usually inorganic (not made by living things).1 For a substance to be classified as a mineral, it must meet five strict criteria:
- Naturally Occurring: It is found in nature, not man-made.
- Inorganic: It did not come from a living organism.2
- Solid: It is not a liquid or a gas.3
- Definite Chemical Composition: It has a specific chemical formula (e.g., Quartz is always 4$SiO_2$).5
- Ordered Internal Structure: Its atoms are arranged in a repeating, geometric pattern called a crystal lattice.6
How Minerals Build Rocks
A rock is simply an aggregate (a collection) of one or more minerals.7 While there are thousands of minerals on Earth, only a few dozen are “rock-forming minerals”—the primary ingredients for most of the Earth’s crust.
1. The Ingredients (Minerals)8
Common minerals like Quartz, Feldspar, and Mica are found in many different types of rocks.9 Because minerals have fixed properties, the specific blend of minerals in a rock determines that rock’s color, hardness, and texture.
2. The Mixing Process (Rock Formation)
The way these mineral “building blocks” are put together determines the type of rock created:
- Igneous Rocks: Formed when molten rock (magma or lava) cools.10 As it cools, different minerals “freeze” and crystallize at different temperatures, interlocking like puzzle pieces.11 Granite is a great example; you can see the distinct pink (feldspar), white (quartz), and black (mica) mineral grains.12
- Sedimentary Rocks: Formed when small mineral grains or pieces of other rocks are pressed and “cemented” together over time.13 In a rock like Sandstone, the building blocks are primarily tiny grains of the mineral quartz.14
- Metamorphic Rocks: Formed when existing rocks are subjected to intense heat and pressure.15 This doesn’t melt the rock but causes the minerals to recrystallize or grow into new patterns, like the bands seen in Gneiss.
Minerals vs. Rocks: A Quick Comparison
| Feature | Mineral | Rock |
| Structure | Uniform (same throughout) | Composite (made of parts) |
| Chemical Formula | Definite (e.g., $NaCl$ for Salt) | Variable (no single formula) |
| Shape | Often forms geometric crystals | Random shapes and sizes |
| Analogy | The individual LEGO bricks | The castle built from the bricks |
It is true that minerals make up rocks?
It is absolutely true. In geology, the simplest way to think of it is: minerals are the ingredients, and rocks are the finished dish.
While we often use the words interchangeably in casual conversation, they have very specific scientific meanings that explain how our planet is built.
1. The Building Block Relationship
A mineral is a pure substance with a specific “recipe” (chemical formula) and a neat, repeating internal structure.1 A rock is what you get when you take one or more of those minerals and lock them together into a solid mass.2
- Monomineralic Rocks: Some rocks are made of just one mineral.3 For example, Limestone is almost entirely made of the mineral Calcite.4
- Polymineralic Rocks: Most rocks are a “stew” of many different minerals. Granite is the classic example—if you look closely at a piece, you can see separate grains of pink feldspar, white quartz, and black mica.5
2. Key Differences at a Glance
Even though they are related, they have different “rules” they have to follow:
| Feature | Mineral (The Ingredient) | Rock (The Mixture) |
| Composition | Fixed and uniform (always the same formula). | Variable (a mix of different things). |
| Structure | Orderly “crystal lattice” atoms. | A random jumble of grains or crystals. |
| Organic Matter | Never (must be inorganic). | Sometimes (e.g., Coal is made of old plants). |
| Example6 | Quartz (7$SiO_2$)8 | Sandstone (mostly quartz grains).9 |
3. How They Get “Glued” Together
Minerals don’t just sit next to each other; they are bonded into rocks through geological processes:10
- Interlocking: In igneous rocks like basalt, minerals grow into each other as they cool from lava, creating a tight “jigsaw” fit.
- Cementation: In sedimentary rocks, minerals like silica or calcite act as a natural glue, filling the gaps between sand grains and hardening into solid stone.11
- Recrystallization: In metamorphic rocks, intense heat and pressure cause the mineral grains to change shape and “knit” together without fully melting.12
Would you like to know which minerals are the most common in the Earth’s crust, or perhaps how geologists tell two similar-looking minerals apart?
Learning about Rocks and Minerals
This video provides a great visual breakdown of how minerals combine to form the different types of rocks we see in the world around us.
What are minerals?
In geology, minerals are the individual, pure substances that make up the solid Earth.1 While people often use “rock” and “mineral” to mean the same thing, they are actually quite different.2 If a rock is a “cookie,” then minerals are the “flour, sugar, and chocolate chips” used to bake it.3
To be officially classified as a mineral, a substance must meet five specific criteria:4
The 5 Criteria of a Mineral
- Naturally Occurring: It must be found in nature (not man-made in a lab).5
- Inorganic: It is not made by living organisms (e.g., wood and pearls are not minerals).6
- Solid: It cannot be a liquid or a gas at standard Earth temperatures.7
- Definite Chemical Composition: It has a specific “recipe” of elements.8 For example, Quartz is always Silicon and Oxygen (9$SiO_2$).10
- Ordered Internal Structure: Its atoms are arranged in a repeating, geometric pattern called a crystal lattice.11
How Geologists Identify Minerals
Because many minerals look similar at first glance, scientists use several physical tests to tell them apart:12
- Hardness: Measured using the Mohs Scale from 1 (softest, like Talc) to 10 (hardest, like Diamond).13
- Luster: How light reflects off the surface (e.g., metallic, glassy, or dull).14
- Streak: The color of the mineral in powder form, found by rubbing it across a ceramic plate.15
- Cleavage: The way a mineral breaks along flat, smooth planes.16
- Color: While helpful, color can be misleading because impurities can change a mineral’s appearance (e.g., Quartz can be clear, purple, or pink).17
Common Examples
- Quartz: The most common mineral in the Earth’s crust; used to make glass.
- Halite: Common table salt (18$NaCl$).19
- Hematite: An iron ore that often leaves a reddish-brown streak.20
- Mica: Known for splitting into thin, flexible, see-through sheets.21
Minerals vs. Rocks: The Main Difference
The biggest difference is that a mineral is homogenous (the same throughout), whereas a rock is an aggregate—a mixture of different minerals.22 If you look closely at a piece of Granite, the different colored “specks” you see are the individual minerals (Quartz, Feldspar, and Mica) that have grown together.23
What is the atomic structure of matter?
To understand the atomic structure of matter, we have to look at the atom, which is the fundamental building block of everything in the universe.1 While atoms are incredibly small—about a million times thinner than a human hair—they are made up of even smaller subatomic particles.2
1. The Core Components
An atom is composed of two distinct regions: a tiny, dense center and a large, mostly empty outer area.3
- The Nucleus (The Center): This is the “heart” of the atom.4 It contains more than 99.9% of the atom’s mass but occupies only a tiny fraction of its volume.5
- Protons: Positively charged (6$+1$) particles.7 The number of protons determines what element the atom is (e.g., every Carbon atom has 6 protons).8
- Neutrons: Particles with no charge (neutral).9 They act like “nuclear glue,” helping to hold the protons together.10
- The Electron Cloud (The Outer Region):
- Electrons: Negatively charged (11$-1$) particles that zip around the nucleus.12 They have almost no mass but determine how the atom bonds with others.
2. Comparing Subatomic Particles
| Particle | Charge | Location | Relative Mass |
| Proton | Positive ($+1$) | Inside Nucleus | 1 |
| Neutron | Neutral ($0$) | Inside Nucleus | 1 |
| Electron13 | Negative (14$-1$)15 | Outside Nucleus16 | ~0 (negligible)17 |
3. How Atoms Stay Together
Atoms are held together by two fundamental forces:
- Electromagnetic Force: Opposites attract.18 The positive charge of the nucleus pulls on the negative charge of the electrons, keeping them from flying away.19
- Strong Nuclear Force: Since like-charges repel, protons in the nucleus naturally want to fly apart.20 The “strong force” overcomes this repulsion, binding protons and neutrons together into a stable core.21
4. Modern vs. Old Models
Our understanding of where electrons live has evolved:
- The Bohr Model (Older): Pictured electrons orbiting the nucleus in neat, circular paths like planets around the sun.22
- The Quantum Mechanical Model (Modern): Suggests electrons exist in a “cloud.”23 We can’t predict their exact path, only the probability of where they might be at any given time.
The Basic Structure of the Atom
This video provides a clear visual breakdown of how protons, neutrons, and electrons are arranged to form the various elements of the universe.
What are chemical reactions?
A chemical reaction is a process where one or more substances (called reactants) are transformed into entirely new substances (called products).1
Think of it like LEGOs: you start with a specific structure, break the pieces apart, and snap them back together in a different way to build something new. The individual “bricks” (atoms) are never destroyed; they are simply rearranged.2
1. How Reactions Work: Breaking and Making Bonds3
For a reaction to happen, the chemical bonds holding the reactants together must be broken.4 This requires energy.5 Once broken, the atoms move around and form new bonds to create the products.6
- Reactants: The starting materials (written on the left side of a chemical equation).7
- Products: The new substances formed (written on the right side).8
- The Arrow (9$\rightarrow$): This represents the change itself, often read as “yields” or “forms.”10
2. Five Signs a Reaction Is Happening11
Since you can’t see atoms moving, geologists and chemists look for “clues” that a chemical change has occurred:12
- Color Change: Like a silver spoon turning black (tarnishing) or a leaf turning red in autumn.
- Temperature Change: The mixture gets significantly hotter (exothermic) or colder (endothermic).13
- Gas Formation: You see bubbles or smell a new odor (like the vinegar and baking soda “volcano”).14
- Precipitate Formation: Two liquids are mixed and a solid “powder” suddenly forms and sinks to the bottom.15
- Light/Energy Production: Like the glow of a firefly or the flame of a candle.16
3. Common Types of Reactions
Most reactions fall into a few basic “playbooks”:
| Type | Description | Simple Analogy |
| Synthesis | Two things combine to make one. | $A + B \rightarrow AB$ |
| Decomposition | One thing breaks down into two. | $AB \rightarrow A + B$ |
| Combustion | A fuel reacts with oxygen to release energy. | Burning wood |
| Replacement | Atoms “swap” partners. | Switching dance partners |
4. The Law of Conservation of Mass
One of the most important rules in science is that matter cannot be created or destroyed.17 This is why chemical equations must be “balanced.” If you start with 4 Hydrogen atoms in your reactants, you must have 4 Hydrogen atoms in your products, even if they are now bonded to something else.
CH4 + 2O2 → CO2 + 2H2O
(In the equation above, notice how the number of Carbon, Hydrogen, and Oxygen atoms is the same on both sides.)
Would you like to see some examples of chemical reactions that happen inside the human body, like digestion or cellular respiration?
Types of Chemical Reactions
This video provides a helpful visual guide to the five major types of chemical reactions, including synthesis and decomposition, with easy-to-follow examples.
What are chemical bonds?
A chemical bond is the physical force of attraction that holds atoms together.1 In our earlier analogy, if atoms are LEGO bricks, chemical bonds are the “studs and tubes” that allow the bricks to snap together and stay connected.
Atoms bond because they want to become stable.2 Most atoms are “unhappy” when they have an incomplete outer shell of electrons.3 By bonding, they can fill those shells—usually aiming for a total of eight electrons, a rule known as the Octet Rule.4
1. The Three Primary Types of Bonds
There are three main ways atoms “shake hands” to form a bond, depending on whether they are metals or non-metals.5
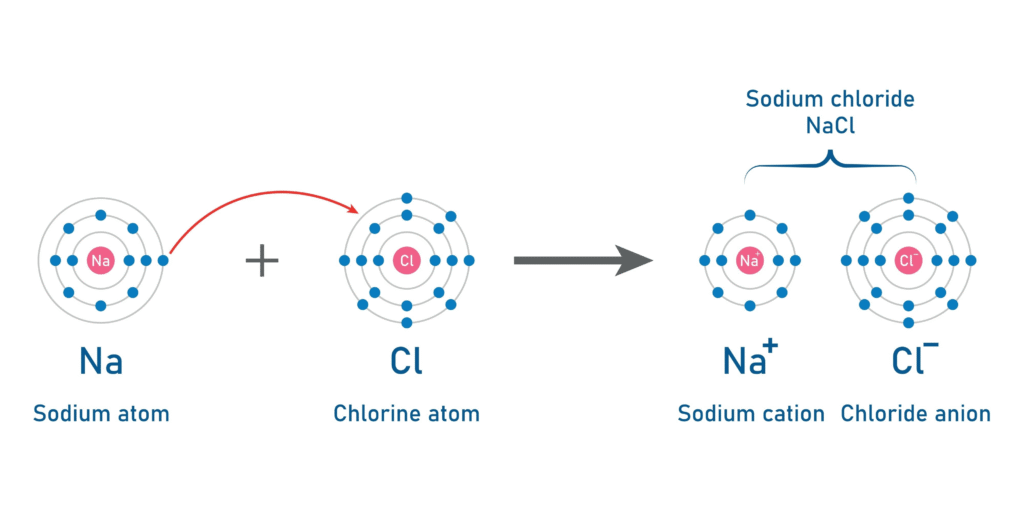
Ionic Bonds: “The Taker”
This happens when one atom completely transfers one or more electrons to another.6 This usually occurs between a metal and a non-metal.7
- How it works: One atom loses an electron and becomes positive; the other gains one and becomes negative.8 Because opposites attract, they stick together tightly.9
- Example: Sodium (10Na) gives an electron to Chlorine (11Cl) to form Table Salt (12NaCl).13
Covalent Bonds: “The Sharers”
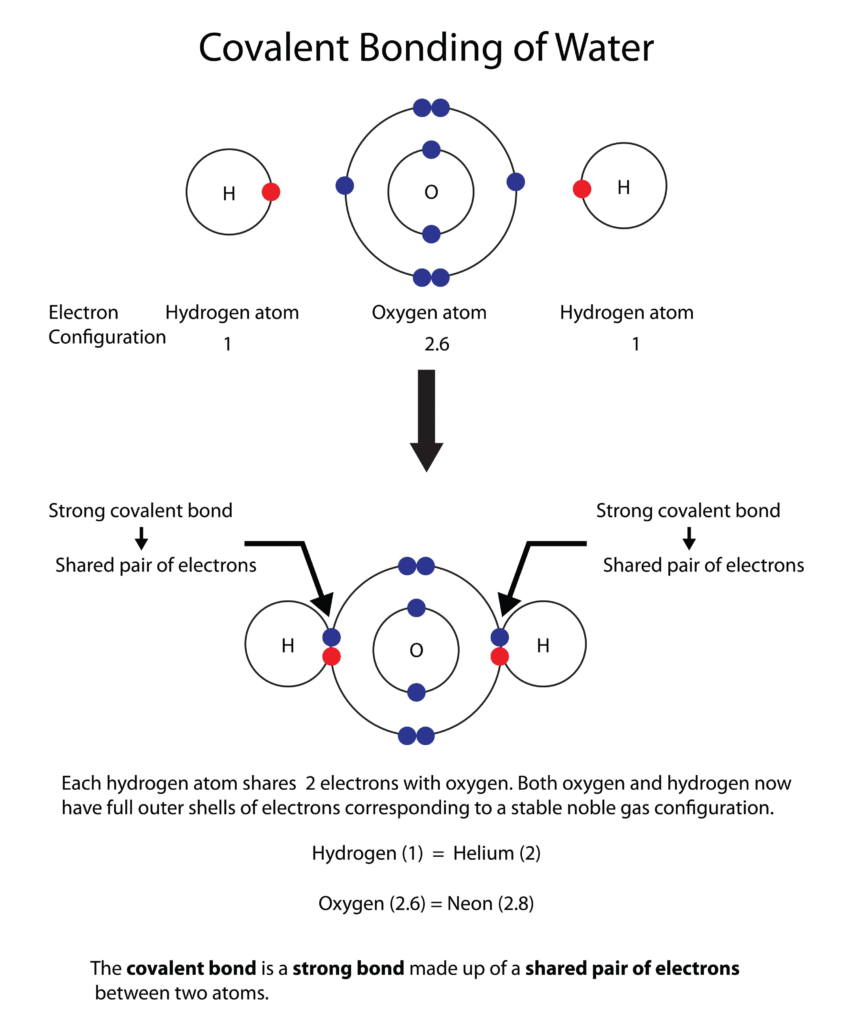
In this bond, atoms share pairs of electrons.14 This is common between two non-metals.
- How it works: Neither atom is strong enough to steal the electron away, so they “co-parent” the electrons to finish their outer shells.15
- Example: Two Hydrogen atoms share electrons with one Oxygen atom to form Water (16H2O).17
Metallic Bonds: “The Community”
Found in pure metals, this bond is like a “sea of electrons.”18
- How it works: Metal atoms drop their outer electrons into a shared pool.19 These electrons flow freely around all the atoms, which is why metals conduct electricity so well.20
- Example: The atoms in a Gold bar or a Copper wire.21
2. Comparing the Bonds
| Feature | Ionic | Covalent | Metallic |
| Electron Action | Transferred | Shared | Delocalized (Shared “Sea”) |
| Types of Atoms | Metal + Non-metal | Non-metal + Non-metal | Metal + Metal |
| Strength | Very High | High to Moderate | Variable |
| Result | Brittle Crystals | Molecules (Gases/Liquids) | Malleable Solids |
3. Why Bonds Matter for Rocks and Minerals
Everything we discussed about minerals earlier comes down to these bonds:
- Hardness: Diamonds are hard because their carbon atoms are held together by extremely strong covalent bonds in every direction.
- Cleavage: Some minerals (like Mica) break in flat sheets because they have strong bonds within the layers but very weak “secondary” bonds between the layers.
- Solubility: Salt ($NaCl$) dissolves in water easily because water molecules can pull apart the ionic bonds holding the salt crystal together.
Would you like to see how these bonds create the specific geometric shapes of crystals?
Chemical Bonding Explained
This video provides a helpful visual guide to the three major types of chemical bonds—ionic, covalent, and metallic—showing how atoms interact to create stable substances.
What is the atomic structure of minerals?
The atomic structure of a mineral refers to the unique, highly organized arrangement of its atoms, ions, or molecules.1 While a rock is a random mixture, a mineral is a masterpiece of geometric precision.
The defining feature of a mineral’s atomic structure is the crystal lattice—a three-dimensional “skeleton” that repeats perfectly in every direction.2
1. The Unit Cell: The “Blueprint”
Every mineral starts with a unit cell. This is the smallest group of atoms that shows the full symmetry and structure of the mineral.3
- Think of it like a single decorative tile. When you lay those tiles side-by-side and top-to-bottom, they create a repeating pattern across an entire floor.
- In a mineral, these unit cells stack by the trillions to build the macroscopic crystal you can actually see and touch.
2. The 7 Crystal Systems
Geologists classify mineral structures into seven fundamental geometric “blueprints.”4 These systems are defined by the lengths of the axes and the angles between them:
| Crystal System | Symmetry Characteristics | Common Example |
| Cubic (Isometric) | 3 equal axes at 90° angles. | Halite (Salt), Diamond |
| Tetragonal | 2 equal axes, 1 different; all at 90°. | Zircon |
| Hexagonal | 3 equal axes at 120°, 1 perpendicular. | Beryl (Emerald), Ice |
| Orthorhombic | 3 unequal axes, all at 90°. | Topaz, Olivine |
| Monoclinic | 3 unequal axes, only 2 at 90°. | Gypsum, Mica |
| Triclinic | 3 unequal axes, none at 90°. | Turquoise |
| Trigonal | Similar to Hexagonal but lower symmetry. | Quartz, Calcite |
3. Polymorphs: Same Atoms, Different Structure5
One of the most fascinating aspects of mineral atomic structure is polymorphism.6 This occurs when two minerals have the exact same chemical formula but different atomic arrangements because they formed under different temperatures or pressures.
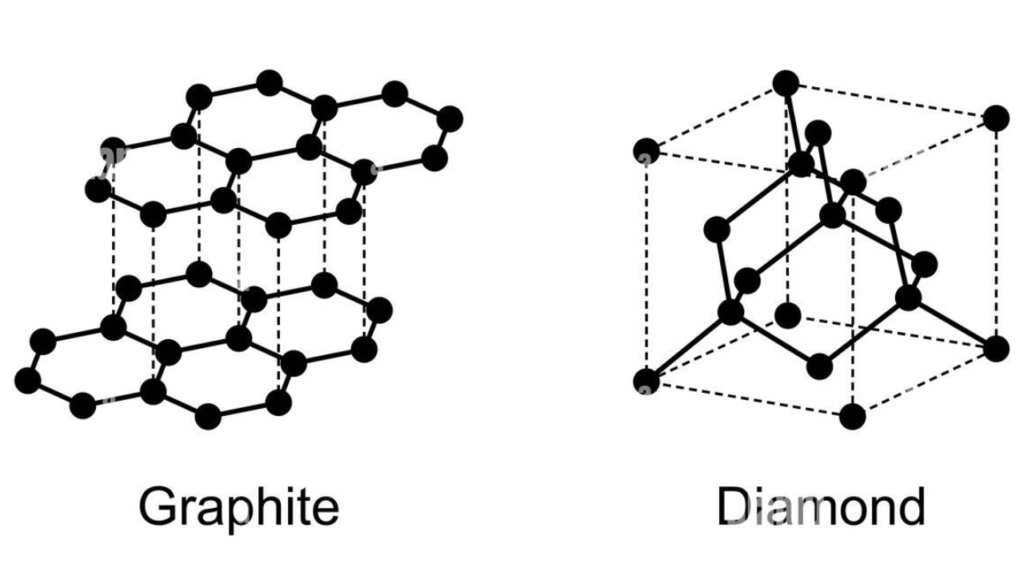
- Diamond vs. Graphite: Both are made of pure Carbon (7$C$).8
- In Diamond, the atoms are locked in a rigid, three-dimensional cubic framework of strong covalent bonds, making it the hardest natural substance.9
- In Graphite, the atoms are arranged in flat, hexagonal sheets.10 While the bonds within the sheets are strong, the bonds between the sheets are incredibly weak, allowing them to slide off—which is why you can write with a pencil.
4. How Structure Dictates Properties
The internal “skeleton” of a mineral determines its physical personality:
- Cleavage: If the atomic bonds are weaker in one direction than another, the mineral will break along that flat plane.11 Mica has a “sheet” structure, so it peels into thin flakes.
- Hardness: Strong, multidirectional bonds (like the covalent bonds in Quartz) result in a harder mineral.12
- Crystal Habit: The outward shape of a crystal (like the perfect cubes of salt) is a direct reflection of its internal cubic lattice.13
What are the rock-forming minerals?
While there are thousands of known minerals on Earth, the vast majority of our planet’s crust is built from just a small handful of common varieties.1 These are known as rock-forming minerals.2
To be classified as a “rock-forming mineral,” a mineral must be abundant in the crust and be one of the original components present when the rock first formed.3
1. The Major Groups
About 90% of the Earth’s crust is made of silicates—minerals that contain a combination of silicon and oxygen.4
The “Big Eight” Rock-Forming Minerals
- Feldspars: The most abundant mineral group in the crust. You’ll find them in almost every igneous rock (like the pink or white parts of granite).
- Quartz: Extremely hard and chemically stable; it’s the primary ingredient in sand and sandstone.5
- Micas: Known for splitting into thin, flexible sheets.6 Common types include Muscovite (clear) and Biotite (black).7
- Amphiboles: Usually dark-colored, needle-like crystals found in rocks like diorite.
- Pyroxenes: Dark green to black minerals that are major components of the oceanic crust (basalt).8
- Olivine: A distinctive olive-green mineral that forms deep in the Earth’s mantle.9
- Calcite: A non-silicate mineral that is the primary “building block” for limestone and marble.10
- Clays: Fine-grained minerals formed from the weathering of other minerals; they make up the bulk of shale and mudstone.11
2. How They Build Different Crusts
The “recipe” of these minerals changes depending on where you are on Earth:
- Continental Crust (Granitic): Mostly made of “Felsic” minerals—Quartz and Feldspar.12 These are lighter in color and less dense.
- Oceanic Crust (Basaltic): Mostly made of “Mafic” minerals—Pyroxene and Plagioclase Feldspar.13 These are darker, heavier, and rich in iron and magnesium.
3. Identification at a Glance
Geologists use specific traits to identify these “building blocks” within a rock:
| Mineral | Key Identification Feature | Common Rock Type |
| Quartz | Glassy luster, no cleavage, very hard. | Granite, Sandstone |
| Feldspar | Rectangular crystals, pearly luster. | Granite, Basalt |
| Calcite | Fizzes when in contact with acid. | Limestone, Marble |
| Biotite | Shiny black “sheets” or flakes. | Schist, Granite |
| Olivine14 | Sugary, olive-green grains.15 | Peridotite, Basalt |
Accessory Minerals
Minerals that appear in very small amounts (like Zircon or Garnet) are called accessory minerals.16 Even though they aren’t the main building blocks, they are like “spices” in the recipe—they help geologists determine the age and history of the rock.17
Would you like to learn how Bowen’s Reaction Series explains the specific order in which these minerals crystallize from cooling magma?
Common Rock-Forming Minerals
This video provides a helpful overview of the most common minerals found in the Earth’s crust and how they are used to classify different rock types.
What are the physical properties of minerals?
Because minerals are solid chemicals with a specific internal structure, they have constant physical properties that geologists use like “fingerprints” to identify them.1
While two minerals might both look like “green rocks,” their physical properties will tell you if one is a piece of glass and the other is a valuable Emerald.
The Primary Identification Properties
1. Hardness
This is a measure of how well a mineral resists being scratched.2 It is measured using the Mohs Hardness Scale, which ranges from 1 (softest) to 10 (hardest).3
- The Test: You try to scratch the unknown mineral with objects of known hardness (like a fingernail, a penny, or a steel nail).4
- Example: If a mineral can scratch a copper penny (Hardness 3) but is scratched by a glass plate (Hardness 5.5), its hardness is about 4 or 5.
2. Luster5
Luster describes how light reflects off the surface of a mineral.6 It’s essentially the “quality of the shine.”
- Metallic: Looks like a polished metal (e.g., Pyrite or Gold).7
- Non-Metallic: Can be described as Vitreous (glassy), Pearly, Silky, Resinous (like sap), or Earthy (dull).8
3. Streak
The streak is the color of a mineral in its powdered form.9
- The Test: Rub the mineral across an unglazed porcelain plate (a “streak plate”).10
- Why it matters: Interestingly, the streak color is often different from the surface color.11 For instance, the mineral Hematite can look silver or black, but it always leaves a reddish-brown streak.12
4. Cleavage and Fracture
These properties describe how a mineral breaks when hit with a hammer.13
- Cleavage: The mineral breaks along smooth, flat planes.14 This happens because the atomic bonds are weaker in certain directions. (e.g., Mica peels into perfect flat sheets).15
- Fracture: The mineral breaks into irregular, jagged, or curved surfaces.16 Quartz famously has “conchoidal fracture,” which looks like the curved ripples inside a broken soda bottle.
Summary of Key Properties
| Property | Definition | Common Example |
| Color | The outward visual hue. | Least reliable (Quartz can be any color). |
| Streak | Color of the mineral’s powder. | Very reliable (Hematite is always reddish-brown). |
| Specific Gravity | How “heavy” it feels for its size. | Gold has a very high specific gravity. |
| Diaphaneity | How much light passes through. | Transparent, Translucent, or Opaque.17 |
“Special” Properties
Some minerals have unique tricks that make them incredibly easy to identify:
- Magnetism: Magnetite will literally stick to a magnet.18
- Effervescence: If you drop a little acid on Calcite, it will fizz and bubble.19
- Taste: Halite (rock salt) tastes salty.20 (Geologists generally advise against tasting unknown minerals, though!)
- Fluorescence: Some minerals, like certain types of Fluorite, glow bright neon colors under a UV light.21
While physical properties (like hardness and color) tell you what a mineral looks like, chemical properties tell you what it is made of and how it reacts with the world around it. These properties are determined by the specific atoms inside the mineral and how they are bonded together.
1. Chemical Composition (The Formula)
Every mineral has a specific chemical “recipe.”1 This is the most fundamental chemical property because it determines all other traits.
- Definite Composition: Minerals like Quartz always have the formula 2SiO2.3
- Variable Composition: Some minerals allow certain atoms to “swap” places.4 In Olivine 5(Mg, Fe)2SiO4, Magnesium and Iron can trade spots depending on the environment where the mineral formed.6
2. Chemical Classification (The Mineral Groups)
Geologists group minerals based on their anion (the negatively charged part of their formula). This is the standard way to organize the thousands of minerals on Earth:
| Mineral Group | Identifying Component | Example |
| Silicates | Silicon and Oxygen (SiO4) | Quartz, Feldspar |
| Carbonates | Carbonate ion (CO32-) | Calcite (fizzes in acid) |
| Oxides | Oxygen (O) + Metal | Hematite (Iron Ore) |
| Sulfides | Sulfur (S) + Metal | Pyrite (Fool’s Gold) |
| Halides | Halogen (Cl, F, Br, etc.) | Halite (Table Salt) |
| Native Elements7 | Single element only8 | Gold, Diamond, Silver9 |
3. Chemical Reactivity
This describes how a mineral behaves when it encounters other chemicals. This is a vital tool for identification in the field.
- Reaction to Acid: This is the “fizz test.” Carbonate minerals like Calcite will bubble (effervesce) when a drop of weak hydrochloric acid is placed on them, releasing 10CO2 gas.11
- Solubility: Some minerals, like Halite, dissolve easily in water.12 Others, like Quartz, are chemically “stubborn” and won’t dissolve even in strong acids.
- Oxidation: Minerals with high iron content, like Pyrite, react with oxygen and water to form rust (Iron Oxide).13 This is why some rock faces look orange or brown.
4. Stability and Weathering
A mineral’s chemical stability determines how long it lasts on the Earth’s surface.
- High Stability: Quartz is very stable; it doesn’t react easily with air or water, which is why it’s the main ingredient in beach sand.14
- Low Stability: Feldspar is less stable.15 Through a chemical process called hydrolysis, it reacts with rainwater to turn into soft Clay.16
Would you like to know more about how chemical weathering actually breaks down these minerals to create soil?
Mineral Groups Explained
This video provides an excellent visual walkthrough of how minerals are categorized into groups like silicates and carbonates based on their chemical formulas.
Solved Problems
1. Problem: Why can’t we classify a pearl or a piece of coal as a mineral, even though they are solid and naturally occurring?
Solution: A mineral must be inorganic. Pearls are created by mollusks (organic) and coal is formed from decomposed plant matter (organic).
2. Problem: Why do atoms form chemical bonds instead of remaining as isolated particles?
Solution: Atoms seek stability. Most atoms have incomplete outer electron shells; by bonding (ionic or covalent), they fill these shells to reach a lower-energy, stable state (the Octet Rule).
3. Problem: If Diamond and Graphite are both made of 100% Carbon, why is one the hardest natural substance while the other is soft enough to write with?
Solution: This is due to Polymorphism. In Diamond, carbon atoms use strong covalent bonds in a rigid 3D lattice. In Graphite, atoms are bonded in flat sheets with very weak van der Waals forces between the layers, allowing them to slide apart.
4. Problem: How does the “Strong Nuclear Force” prevent an atom from flying apart?
Solution: Protons are all positively charged and naturally repel each other. The Strong Nuclear Force acts like “nuclear glue” to overcome this electromagnetic repulsion, holding the protons and neutrons together in the nucleus.
5. Problem: Why are metallic bonds responsible for the electrical conductivity of certain minerals?
Solution: Metallic bonds create a “sea of electrons” where electrons are not tied to one atom but flow freely. This mobility allows electrical current to pass through the material easily.
Mineral Properties & Identification
6. Problem: A geologist finds a clear mineral. It can be scratched by a steel nail but not by a copper penny. What is its approximate hardness on the Mohs scale?
Solution: A penny is 3 and a steel nail is about 5.5. Therefore, the mineral has a Mohs hardness of approximately 4.
7. Problem: Why is “color” considered the least reliable physical property for identifying minerals like Quartz?
Solution: Trace amounts of chemical impurities can drastically change a mineral’s color without changing its structure (e.g., iron turns clear quartz into purple Amethyst).
8. Problem: What chemical property allows a geologist to instantly distinguish Calcite from Quartz?
Solution: Effervescence. Calcite is a carbonate ($CaCO_3$) and will fizz when it reacts with weak hydrochloric acid; Quartz ($SiO_2$) will not.
9. Problem: Why does the mineral Mica break into thin, flat sheets (perfect cleavage) while Quartz breaks in curved, jagged shapes (fracture)?
Solution: Mica has an atomic structure with weak bond planes in one direction. Quartz has strong covalent bonds that are equally strong in all directions, so it has no “weak” path to follow when it breaks.
10. Problem: If a mineral has a “Metallic Luster” but leaves a “Dark Gray Streak,” is it more likely to be Graphite or Hematite?
Solution: Graphite. Hematite is famous for leaving a distinct reddish-brown streak, regardless of how metallic or black the surface looks.
Rock-Forming Processes
11. Problem: Why are Silicate minerals ($SiO_4$) the most common rock-forming minerals in the Earth’s crust?
Solution: Silicon and Oxygen are the two most abundant elements in the Earth’s crust by weight. They easily bond to form the silicon-oxygen tetrahedron, the “building block” of most rocks.
12. Problem: How does the cooling rate of magma affect the size of mineral crystals in an igneous rock?
Solution: Slow cooling (underground) allows atoms more time to migrate to a crystal lattice, creating large crystals. Fast cooling (surface) results in tiny or microscopic crystals.
13. Problem: Why is Oceanic Crust denser than Continental Crust?
Solution: Oceanic crust is made of Mafic minerals (rich in Iron and Magnesium), which are naturally heavier. Continental crust is made of Felsic minerals (Quartz/Feldspar), which are rich in lighter elements like Aluminum and Silicon.
14. Problem: What role does “Cementation” play in the formation of sedimentary rocks?
Solution: Cementation is a chemical process where minerals (like Calcite or Silica) precipitate out of water and fill the spaces between sediment grains, “gluing” them into a solid rock.
15. Problem: Why do metamorphic rocks often show “foliation” (layering or banding)?
Solution: Under intense directed pressure, platy minerals like Mica are forced to rotate and grow perpendicular to the pressure, creating a layered or striped appearance.
Advanced Mineral Chemistry
16. Problem: How can the same mineral, Olivine, have a variable chemical formula like (Mg, Fe)2SiO4?
Solution: This is Ionic Substitution. Because Magnesium (Mg2+) and Iron (Fe2+) have similar sizes and the same charge, they can substitute for each other in the crystal lattice without changing the mineral’s structure.
17. Problem: What is the difference between a “Rock” and an “Ore”?
Solution: A rock is a general aggregate of minerals. An ore is a specific type of rock that contains a high enough concentration of a valuable mineral (like Gold or Iron) to be mined for a profit.
18. Problem: Why does Feldspar eventually turn into Clay at the Earth’s surface?
Solution: Hydrolysis. This is a chemical weathering process where the hydrogen ions in rainwater react with the mineral grains of Feldspar, breaking them down into soft, fine-grained clay minerals.
19. Problem: What is the “Law of Conservation of Mass” in the context of a chemical reaction?
Solution: It states that matter is neither created nor destroyed. In a chemical reaction, the number of atoms in the reactants must exactly equal the number of atoms in the products.
20. Problem: How does the “Unit Cell” of a mineral relate to its macroscopic crystal shape?
Solution: The crystal habit (outer shape) is a direct geometric reflection of the internal unit cell. For example, Halite has a cubic unit cell, which is why it grows into perfect cubic crystals.
Learn how we bridge these gaps: [The Starline Philosophy: The Modern Polymath]
